
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Sourav Ghosh, Monit Paul, Anusree Raha, Prosenjit Mukherjee, Anindya Bagchi*, Abhik Si
Netaji Subhas Chandra Bose institute of Pharmacy, Chakdaha, Nadia, West Bengal India
*Address for Corresponding Author
Anindya Bagchi
Assistant Professor
Netaji Subhas Chandra Bose Institute of Pharmacy, West Bengal, India
Abstract
Objective: A simple comparative study of evaluation of biochemical oxygen demand (BOD) of river water of Ganga’s has been developed with statistical analysis between far water and near water. Material and Methods: The process has been conducted by simple titrimetric method by taking four 200 ml BOD bottles (two for sample & two for blank) and analysed two different intervals in the gap between five days. The total process was repeated for four times times, one for near water & another for far water. Results: Far water BOD value came less than near water BOD value as the experiment have been performed. Conclusion: So it can be concluded that far water system in more compatible for aquatic life.
Keyword: Biochemical oxygen demand, Ganga’s, river, sodium thiosulphate
Introduction
The biochemical oxygen demand determination is a chemical procedure for determining the amount of dissolved oxygen needed by aerobic organisms in a water body to break the organic materials present in the given water sample at certain temperature over a specific period of time (Assumuth and Strandberg, 1993).
Biochemical oxygen demand (BOD) of water or polluted water is the amount of oxygen required for the biological decomposition of dissolved organic matter to occur under standard condition at a standardized time and temperature. Usually, the time is taken as 5 days and the temperature is 20°C.
The test measures the molecular oxygen utilized during a specified incubation period for the biochemical degradation of organic material (carbonaceous demand) and the oxygen used to oxidize inorganic material such as sulfides and ferrous ion. It also may measure the amount of oxygen used to oxidize reduced forms of nitrogen (nitrogenous demand).
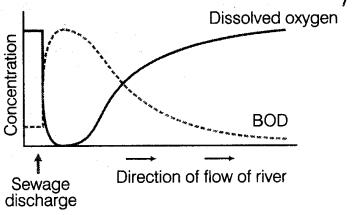 Figure 1. BOD Content variation within river
Figure 1. BOD Content variation within river
BOD is the principle test to give an idea of the biodegradability of any sample and strength of the waste. Hence the amount of pollution can be easily measured by it. Efficiency of any treatment plant can be judged by considering influent BOD and the effluent BOD and so also the organic loading on the unit (Bellos et al., 2004).
Application of the test to organic waste discharges allows calculation of the effect of the discharges on the oxygen resources of the receiving water. Data from BOD tests are used for the development of engineering criteria for the design of wastewater treatment plants. Ordinary domestic sewage may have a BOD of 200 mg/l. Any effluent to be discharged into natural bodies of water should have BOD less than 30 mg/l.
This is important parameter to assess the pollution of surface waters and ground waters where contamination occurred due to disposal of domestic and industrial effluents. Drinking water usually has a BOD of less than 1 mg/l. But, when BOD value reaches 5 mg/l, the water is doubtful in purity. The determination of BOD is used in studies to measure the self-purification capacity of streams and serves regulatory authorities as a means of checking on the quality of effluents discharged to stream waters. The determination of the BOD of wastes is useful in the design of treatment facilities. It is the only parameter, to give an idea of the biodegradability of any sample and self-purification capacity of rivers and streams. The BOD test is among the most important method in sanitary analysis to determine the polluting power, or strength of sewage, industrial wastes or polluted water. It serves as a measure of the amount of clean diluting water required for the successful disposal of sewage by dilution (Assumuth and Strandberg, 1993).
On the basis of the BOD values, the characteristics of the water and the biological activity of the incubated microflora can be determined. Effluent with high BOD levels is discharged into a stream or river; it will accelerate bacterial growth in the river and consume the oxygen levels in the river. The oxygen may diminish to levels that are lethal for most fish and many aquatic insects. As the river re-aerates due to atmospheric mixing and as algal photosynthesis adds oxygen to the water, the oxygen levels will slowly increase downstream. The biological capacity of a sewage treatment plant can be tested by comparing the BOD value of a known control solution with the BOD derived from the treatment plant.
BOD detects only the destructible proportion of organic substances and as a general principle is therefore lower than the COD value, which also includes inorganic materials and those materials which cannot be biologically oxidized (Assumuth et al., 1993; Bellos et al., 2004).
Materials and Methods
Principle
The sample is filled in an airtight bottle and incubated at specific temperature for 5 days. The dissolved oxygen (D.O) content of the sample is determined before and after five days of incubation at 20°C and the BOD is calculated from the difference between initial and final D.O. The initial DO is determined shortly after the dilution is made; all oxygen up take occurring after this measurement is included in the BOD measurement.
Chemicals and reagents
Calcium Chloride, Magnesium Sulphate, Ferric Chloride and Potassium Di Hydrogen Phosphate were purchased from Merck India Pvt. Ltd. also Disodium hydrogen phosphate, Ammonium Chloride, Manganese sulphate, Potassium hydroxide, Potassium iodide and Sodium azide were purchased from Loba Chem India Pvt. Ltd.
Determination of biochemical oxygen demand
Simple titrimetric method has been followed in the analysis part and BOD incubator was used for the preservation of test sample. Titrate has been carried out with standard sodium thiosulphate solution with the sample solution until the yellow color of liberated Iodine is almost faded out (Pale yellow color). Add 1 ml of starch solution and continue the titration until the blue color disappears to colourless with the identification of the volume of sodium thiosulphate solution consumed, which gives the D.O. in mg/l.
All the data has been statistically validated (Mahajan et al., 2005).
Results and discussion
For determining the Biochemical Oxygen Demand in the given Ganga’s water sample, the readings should be tabulated.
Table 1. BOD of near water
|
Vol. of sample |
Vol. of dil. sample |
Trial No: |
VC(DO)
|
VC(D5)
|
VC(BL0) Blank initial |
VC(BL5) Blank after 5 days |
BC
|
o2 demand
|
o2 demand final |
|
|
10 |
200 |
1 |
9.6 |
5.2 |
4.3 |
4 |
0.3 |
820 |
82 |
|
|
|
|
2 |
9.3 |
4.9 |
4.4 |
4.2 |
0.2 |
840 |
84 |
|
|
|
|
3 |
9.5 |
5 |
4.4 |
4.1 |
0.3 |
840 |
82 |
|
|
|
|
4 |
9.3 |
5.1 |
4.5 |
4.4 |
0.1 |
820 |
82 |
|
|
Mean SD % RSD |
83 1.154700538 1.391205468 |
|||||||||
We have repeated the method 4 times & the method was found system precise.
Calculation
Initial DO of the diluted sample, Do
DO at the end of the 5 days for the diluted sample, D5
Blank correction =Co – C5, BC
Initial DO of the blank, Co
DO at the end of 5 days for the blank, C5
Biochemical Oxygen Demand
= (D0 – D5 –BC)x Volume of the diluted sample/ Volume of sample taken
Table 2. BOD of far water
|
Vol of sample
|
Vol of dil sample |
Trial No:
|
VC(DO)
|
VC(D5)
|
VC(BL0) Blank initial |
VC(BL5 Blank after 5 days |
BC
|
o2 demand
|
o2 demand final |
|
10
|
200
|
1 |
8.8 |
4.7 |
4.1 |
3.9 |
0.2 |
780 |
78 |
|
2 |
8.5 |
4.9 |
4.2 |
3.8 |
0.4 |
640 |
64 |
||
|
3 |
8.7 |
4.8 |
3.9 |
3.5 |
0.4 |
700 |
70 |
||
|
4 |
8.7 |
4.6 |
3.8 |
3.6 |
0.2 |
780 |
78 |
||
|
Mean |
72.5 |
||||||||
|
SD |
6.806859286 |
||||||||
|
% RSD |
9.388771428 |
||||||||
Experiment has been repeated the method 4 times & the method was found to be not system precised.
Initial DO of the diluted sample, Do
DO at the end of the 5 days for the diluted sample, D5
Blank correction =Co – C5, BC
Initial DO of the blank, Co
DO at the end of 5 days for the blank, C5
Biochemical Oxygen Demand:
= (D0 – D5 –BC) x Volume of the diluted sample/ Volume of sample taken
Table 3. O2 demand comparison b/w near & far water
|
Trial No. |
Near |
Far |
Mean |
SD |
% RSD |
|
1 |
82 |
78 |
80 |
2.82843 |
3.535534 |
|
2 |
84 |
64 |
74 |
14.1421 |
19.11099 |
|
3 |
84 |
70 |
77 |
9.89949 |
12.85649 |
|
4 |
82 |
78 |
80 |
2.82843 |
3.535534 |
We have also compared two sources of water with each other 4 times but not a single trial has given us precise result.
Discussion
Thus, the mean value of biochemical oxygen demand of the Near Ganga’s water is 83 mg/L & Far Ganga’s water is 72.5 mg/L. There is a difference between BOD of near water & the BOD of the far water as the difference of the clearance of near water & far water. The water flow speed of far water is more than the near water, that’s why far water contained less amount of the bacteria. Therefore BOD level of the far water is more than the near water.
Conclusion
Thus, the biochemical oxygen demand of the Near Ganga’s water is 83 mg/l & Far Ganga’s water is 72.5 mg/l. That is why the aquatic ecosystem can sustain well in the far water rather than in the near water.
Acknowledgement
The authors are thankful to the respected Principal Sir, Dr. Arnab Samanta, Netaji Subhas Chandra Bose Institute of Pharmacy, West Bengal for providing necessary facilities for the completion of research work.
References
Assumuth TW, Strandberg T. 1993 Ground-water contamination at Finnish landfills. Water, Air, & Soil Pollution, 69:179,
Bellos D, Sawidus T, Tsekos I. 2004. Nutrient Chemistry of River Pinios (Thessalia, Greece), Environment International, 30:105-115.
Mahajan SW, Khare S, Shrivastavn VS. 2005. A Correlation and Regression Study, Indian Journal of Environmental Protection, 25(3):254-259.