
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Megha Chouhan, Rahul Saxena*, Harsh Patidar
Department of Pharmacology, Ravishankar College of Pharmacy, Bhopal (M.P.), India
*Address for Corresponding author
Rahul Saxena
Assistant Professor
Department of Pharmacology
Ravishankar College of Pharmacy, Bypass Road, Bhanpur Square, Bhopal (MP), India
Abstract
Objective: Aim of the present study was to evaluate the anti-diarrheal & anti-ulcer activity of hydro-alcoholic extract of aerial part of Tridax procumbens in experimental rats. Material and methods: Adult Wistar rats of 150-200 gm were used for the study. The antidiarrheal activity of Tridax procumbens extract was evaluated by castor oil and magnesium sulfate induced diarrhea using parameters such as onset of diarrhea, total number of stool and weight of total number of stools. The antiulcer activity of extracts was investigated using ethanol and induced ulcer. The gastric content was collected to determine its volume, pH, total acidity and free acidity. The mucosal surface of the stomach was scored in terms of ulcer index. The effect of the extract in these models was comparable to the standard drugs (Loperamide 3 mg/kg and Ranitidine 50 mg/kg) used. Results: Tridax procumbens extract was more efficacious in reducing number of total stools and weight of stools and significantly increase onset of diarrhea in both the models. % Inhibition of defecation of test extract was also found to 44.92% and 55.88% relatively compare to standard 76.81% and 78.97% in both models. The antiulcer activity of the extracts was confirmed by a reduction in ulcer index along with the decrease in gastric volume, total acidity, and an increase in pH of gastric content in both the models. Conclusion: In conclusion, we can say that extract dose (300 mg/kg) of Tridax procumbens show significantly reducing the condition of diarrhea as well as an ulcer with the help of flavonoids, saponin, alkaloid, tannins, phenols and some other phytoconstituents.
Keywords: Diarrhea, Inflammatory diseases, Gastric ulcer, Gastric content, Hemorrhage, Tridax procumbens
Introduction
Diarrhea can be defined as an alteration in the normal bowel movement, characterized by a situation in which an adult daily stools exceeds 300 g and contains 60 – 95 % water. Diarrhea can cause severe dehydration that can lead to death. It represents the second most common cause of infant mortality after pneumonia in developing countries, and it is responsible for the death of about 2 - 4 million children every year, especially in Africa (Wansi et al., 2014). Because of this, the WHO has initiated a Diarrheal Disease Control Program to study traditional medical practices and other related aspects. The major causative agents of diarrhea in humans include Shigella flexneri, Staphylococcus aureus, Escherichia coli, Salmonella typhi, and Candida albicans (Ashrafuzzaman et al., 2016). Based on the duration, diarrhea is classified into 3 types: Acute diarrhea (duration < 2weeks), Persistent diarrhea (duration from 2 to 4 weeks), and Chronic diarrhea (more than 4 weeks) (Mekonnen et al., 2018). Inflammatory diseases cause diarrhea with exudative, secretory or osmotic components. Altered motility of the intestine or colon may alter fluid absorption by increasing or decreasing the exposure of luminal content to the intestinal absorptive surface. However, from a pathophysiologic perspective, no single cause of diarrhea is truly unifactorial (Seth S and MD 2012). Gastric ulcer is a break in the tissue lining of the stomach. Most ulcers can be cured without complications; however, in some cases peptic ulcers can develop, such as in penetration, perforation, bleeding (hemorrhage), and obstruction (Sathish et al., 2011). Gastric ulcer is a major disease of the gastrointestinal system which affects 10% of the world population with different etiologies. Chronic alcohol consumption, smoking, stress, usage of non-steroidal anti-inflammatory drugs and H. pylori infection are the causes of gastric ulcer characterized by inflammation and mucosal bleeding in long-term untreated patients. Excess acid secretion and reduced biosynthesis of prostaglandin E2 are important in gastric ulcer formation (Panneerselvam S, 2011; Vimala et al., 2014). Nature has been a source of medicinal agents for thousands of years and impressive numbers of modern drugs have been isolated from natural resources. Tridax procumbens Linn (Family –Asteraceae) is a common herb found in India, commonly known as coat buttons or tridax daisy. It is best known as widespread weed and pest plant Tridax procumbens is well known Ayurvedic, Ethno-botanical and Unani medicinal plant. It has been extensively used in Indian traditional medicine for wound healing, as an anticoagulant, antifungal, and insect repellent in diarrhea and dysentery. It is also dispensed as ‘Bhringraj’ which is well known ayurvedic medicine for liver disorders. [9] The chemical constituents present are alkaloids, carotenoids, flavonoids (catechins and flavones), saponins and tannins. Mineral composition present in leaves is calcium, magnesium, potassium, sodium, and selenium. Whereas the oleanolic acid, fumaric acid, fl-sitosterol, and tannin is present in good amounts (Agrawal et al., 2010; Mir et al., 2017; Kethamakka et al., 2014). Loperamide (2 mg capsule) is the generic form of the brand-name drug (Imodium), a medication used to treat diarrhea. It is used to relieve the symptoms of diarrhea that start suddenly and lasts for a few days. It is also used to treat long-term diarrhea associated with other diseases (healthline.com; mayoclinic.org/; niddk.nih.gov). Ranitidine (Zinetac150) is a competitive, reversible inhibitor of the action of histamine at the histamine H2 - receptors, including receptors on the gastric cells (dailymed.nlm.nih.gov; drugbank.ca; drugs.com)
Materials and Methods
Material and reagents: Ranitidine (Zinetac150) procured from GSK and Loperamide (Imodium) procured from Janssen pharmaceuticals. Ethanol obtained from Himedia Laboratories Mumbai, India. Chloroform obtained from LOBA Chemie (P) Ltd, Mumbai India. CMC obtained from central drug house (P) LTD, New Delhi India. All other chemicals used in the study were purchased from Rankem Gurgaon, Haryana, India. All reagents were of analytical grade.
Collection and Authentication of plant
The aerial part was collected in July from the local market. Herbarium file of plant part was prepared and authenticated by Dr. Zia Ul Hasan (Professor & Head) Department of Botany, Saifia College Bhopal.
Drying and size reduction of plant material
The aerial part was dried under shade. It was pulverized to a coarse powder with the help of a mixer grinder. The coarse powder was passed through sieve No. 20 to maintain uniformity and packed into an airtight container and stored in a cool and dry place. This material was used for further study.
Preparation of Tridax procumbens aerial part extract
Extraction of aerial part of Tridax procumbens was done by the Soxhlet extraction method. Soxhlet apparatus (Khera) was used for the solvent extraction and hydro-alcohol (1:1) was selected as a solvent for extraction. 500 gm of coarsely dried powder was extracted with hydro-alcohol using soxhlet apparatus for 72 hrs at 60-700C. After complete extraction, the extract was collected & dried at room temperature. The percentage yield of extract was calculated & dried crude extract was stored in an airtight container for further study.
Preliminary phytochemical Analysis
The extract obtained by solvent extraction was subjected to various qualitative tests to detect the presence of common chemical constituents (Khandelwal KR 2003; Kokate CK 1994).
Acute Oral Toxicity Studies (OECD 425): The acute toxic category method is a method for assessing acute oral toxicity that involves the identification of a dose level that causes mortality. Female albino rats of weighing 150-200 g were used for the study. These were acclimatized to laboratory conditions for one week before the start of dosing. The doses were selected according to the OECD guideline no. 425. Two group of healthy rats (each group having 3 rats) were used for this experiment. Animals received a single dose of 2000 mg/kg, per oral was selected for the test, as the test item was a source from an herb. After administration of the extract, food was withheld for 3-4 hrs. Once daily cage-side observations included changes in the skin, fur, eyes, mucous membrane (nasal), autonomic (salivation, lacrimation, urinary incontinence, and defecation), and central nervous system (drowsiness, gait, tremors, and convulsions) changes. Mortality, if any, was determined over 2 weeks (OECD guideline 425).
Experimental Work (CPCSEA Guideline; Vogel Gerhard, 2008)
Animals: Adult Wistar rats of 150-200 g were used for the study. The rats were obtained from the Ravishankar College of Pharmacy Bhopal, (M.P.) for experimental purposes. The animals were maintained under controlled conditions of temperature (23 ± 2°C), humidity (50 ±5%) and 12 h light-dark cycles. All the animals were acclimatized for seven days before the study. The animals were randomized into experimental and control groups and housed individually in sanitized polypropylene cages containing sterile husk as bedding. They had free assessed to standard pellets as basal diet and water ad libitum. All the studies conducted were approved by the Institutional Animal Ethical Committee (IAEC) of Ravishankar college of pharmacy Bhopal, (M.P.) (Proposal no: RCOP/ IAEC/MAY 2018/04) according to prescribed guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animal, Govt. of India. (Approval no. 1733/PO/Ere/S/13/CPCSEA)
Preparation of dose: The hydro-alcoholic extract of Tridax procumbens aerial part was dissolved in the suspending agent (1% CMC) before orally administered to the Rats. Standard drugs were dissolved in the suspending agent (1% CMC) before orally administered to the Rats.
In vivo antidiarrhoeal activity (Tadesse et al., 2014; Kaur et al., 2014):
Castor oil induced diarrhea: Rats of either sex were fasted for 18 h and divided into four groups.
|
Group |
|
|
Normal control |
Received 10% Tween 80 (5 ml/kg) by gavage |
|
isease control |
Received castor oil (1 ml) |
|
Standard control |
Received loparamide (3 mg/kg) + castor oil (1 ml) |
|
Test control |
Received extract (300 mg/kg) + castor oil (1 ml) |
After 30 min, diarrhea was induced by oral administration of castor oil (1 ml) to each rat. Rats were observed for a period of 4 h during which the total number of fecal outputs and the number of diarrheic feces excreted were recorded.
Magnesium sulfate induced diarrhea: Rats of either sex were fasted for 18 h and divided into four groups.
|
Group |
|
|
Normal control |
Received 10% Tween 80 (5 ml/kg) by gavage |
|
Disease control |
Received magnesium sulfate (2 mg/kg) |
|
Standard control |
Received loparamide (3 mg/kg) + magnesium sulfate (2 mg/kg) |
|
Test control |
Received extract (300 mg/kg) + magnesium sulfate (2 mg/kg) |
After 30 min, diarrhea was induced by oral administration of magnesium sulfate (2 mg/kg) to each rat. Rats were observed for a period of 4 hr during which the total number of fecal outputs and the number of diarrheic feces excreted were recorded.
Evaluation: Every hour, the total weight of fecal output, the total weight of wet feces, the total number of fecal output, and the number of wet feces were recorded. A numerical score based on stool consistency was assigned as follows: normal stool = 1, semi-solid stool = 2 and watery stool = 3. And % inhibition of diarrhea was calculated as follows:
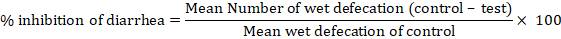
In vivo antiulcer activity (Tadesse et al., 2014; Kaur et al., 2014)
Ethanol induced ulcer: Rats of either sex were fasted for 18 h and divided into four groups.
|
Normal control |
Received 0.9% Nacl (5 ml/kg) by gavage |
|
Disease control |
Received ethanol (1 ml) |
|
Standard control |
Received Ranitidine (50 mg/kg) + ethanol (1 ml) |
|
Test control |
Received extract (300 mg/kg) + ethanol (1 ml) |
After 30 min of respective treatment, ulcer was induced by oral administration of ethanol (1 ml) except in Group I. One hour post ethanol administration, animals were sacrificed and their stomach was removed to examine extent of mucosal damage in terms of ulcer index.
NSAIDS induced ulcer: Rats of either sex were fasted for 18 h and divided into four groups.
|
Normal control |
Received 0.9% Nacl (5 ml/kg) by gavage |
|
Disease control |
Received diclofenac sodium (50 mg/kg) |
|
Standard control |
Received Ranitidine (50 mg/kg) + diclofenac sodium (50 mg/kg) |
|
Test control |
Received extract (300 mg/kg) + diclofenac sodium (50 mg/kg) |
After 30 min of respective treatment, ulcer was induced by oral administration of diclofenac sodium (50 mg/kg) except in Group I. One hour post diclofenac sodium (50 mg/kg) administration, animals were sacrificed and their stomach was removed to examine extent of mucosal damage in terms of ulcer index.
Evaluation parameter (Ingale et al., 2016).
The gastric content was collected to determine its volume, pH, total acidity and free acidity. The mucosal surface of the stomach was scored in terms of ulcer index.
Estimation of pH and gastric volume: The contents of the stomach obtained, was centrifuged at the speed of 1000 revolutions per minute for 20 minutes. The supernatant fluid obtained was collected and its volume was estimated. The pH of the content was noted using pH strips by matching the color obtained with that of the reference standard.
Estimation of free and total acidity: The gastric content obtained was diluted to 10 ml by adding distilled water and in a conical flask. This fluid was analyzed for its free and total acidity by titrating it against 0.01 N NaOH solutions. The initial recording of the fluid level (A) in the burette was noted. 1 to 2 drops of Topfers reagent (dimethylamino azobenzene in 95% alcohol) were added to the conical flask contents and titration was done by constant mixing with the contents of the conical flask. The level at which the color change from red to yellowish orange occurred, that corresponded to the free acidity, was noted (B). The volume of NaOH consumed (B-A) was recorded.
Free acidity was calculated by the formula
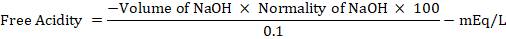
The titration was continued by adding phenolphthalein indicator to the conical flask contents and the level at which the color change from yellowish orange to slight pink occurred was noted. This corresponded to the total acidity and the value was recorded as (C). The volume consumed (C-A) was noted.
Total acidity was calculated by the formula
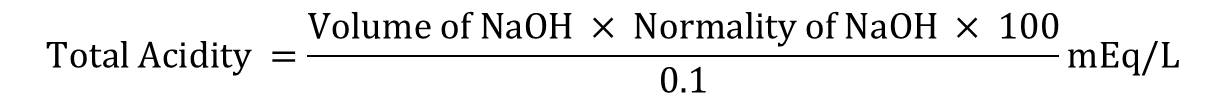
Free acidity and Total acidity were expressed in mEq/litre/100 gm.
Estimation of ulcer number: The gastric mucosa was observed macroscopically under 10x magnifications. The number of superficial ulcers, deep ulcers and perforations were noted. The total number of ulcers in each rat was recorded.
Estimation of gastric lesion: Gastric lesion of all possible ulcers was recorded by measuring its longest diameter (in mm) employing divider and graph paper. The total gastric lesion in each rat was calculated.
Estimation of ulcer severity: The ulcers in each rat were graded as:
0 - No ulcers
1 - Superficial ulcers
2 - Deep ulcers
3 - Perforations
The average severity score of each group was calculated. The mean ulcer score for each animal will be expressed as the ulcer index. The percentage of ulcer protection was determined as follows:-
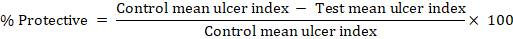
Estimation of ulcer index: The total mucosal area and total ulcerated area were measured. The ulcer index was then calculated using the following equation:
Ulcer index = 10/x
where x is the total mucosal area/ulcerated area
Statistical analysis: The data were expressed as mean ± SEM. Results were analyzed statistically by One-way ANOVA (analysis of variance) followed by Dunnett’s t-test using standard statistical software. All the groups were compared with the control group in each model. The difference was considered significant if p<0.05 (Ingale et al., 2016).
Histopathological evaluation: After the standard processing, the ulcerated gastric tissues were fixed in neutral buffered formalin for 24 h and examined under the microscope for histopathological changes such as inflammation, infiltration, and erosion.
Results
Percentage yield: The percentage yield of aerial part of Tridax procumbens was obtained as 13.25 % (w/w) on a dry basis.
Phytochemical screening: There is a presence of alkaloids, carbohydrates, flavonoids, glycosides, proteins, and saponins in a hydroalcoholic extract of Tridax procumbens aerial part. [Table 1]
Acute Toxicity Studies (LD50): In both phase I and Phase II procedures, none of the animal mortal or any signs of behavioral changes or show any toxicity upon the single administration of HATP (2000 mg/kg per oral). Thus, 300 mg/kg dose was selected for the present study. [Table 2]
Evaluation parameters: Tridax procumbens extract was more efficacious in reducing number of total stools and weight of stools and significantly increase onset of diarrhea in both the models of diarrhea. % Inhibition of defecation of test extract was also found to 44.92% and 55.88% relatively compare to standard 76.81% and 78.97% in both models. The antiulcer activity of the extracts was confirmed by a reduction in ulcer index along with the decrease in gastric volume, total acidity, and an increase in pH of gastric content in both the models. [Table 3, 4, 5, 6]
Morphological study: In a normal group, stomach integrity was maintained and appeared normal. In the disease control group, severe bleeding, perforation, spot ulcer, streaks were observed. The gastric mucosa was appeared intact in the stomach of ranitidine treated animals. The gastric mucosa was seen to be slightly damaged with red coloration, spot ulcers and a few hemorrhagic streaks in the stomach of test control (300 mg/kg) treated animals. [Figure 1]
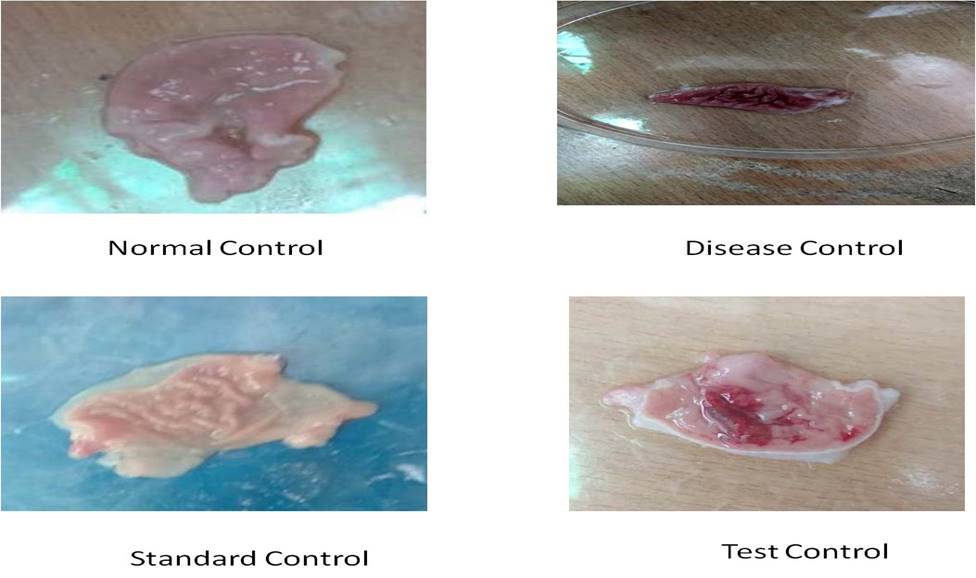
Figure 1. Morphological features of stomach during ulceration study
Histopathological study: Mucosal organization was normal in normal control animals. In the disease control group, gastric mucosa showed many pit‑shaped ulcers, mucosal and submucosal congestion, along with surface erosion at several places. In the ranitidine treated group, gastric mucosa revealed mild superficial erosions with intact submucosa and deeper mucosa. In test control (300 mg/kg) groups, superficial erosions and a few ulcers with mild disorganization of mucosa were observed. [Figure 2 and 3]
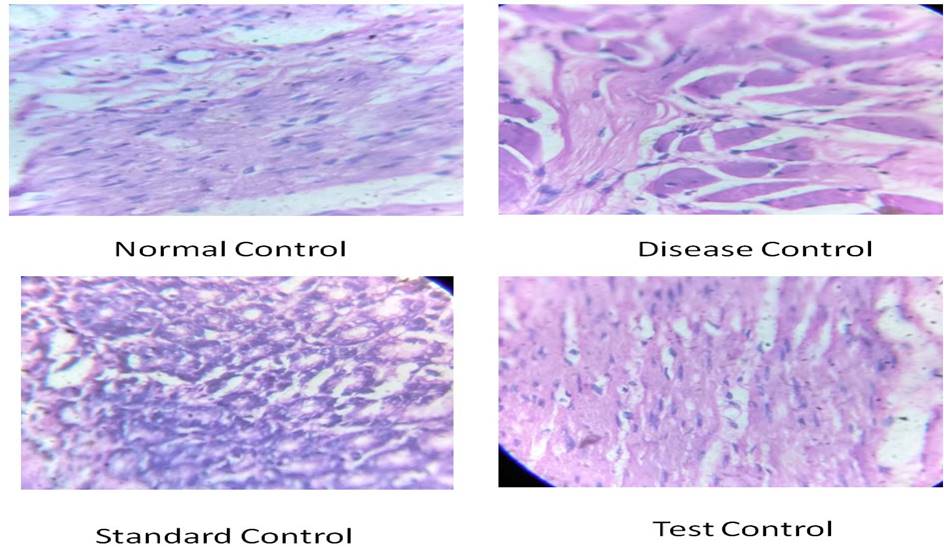
Figure 2. Histological study of stomach in ethanol induced anti-ulcer
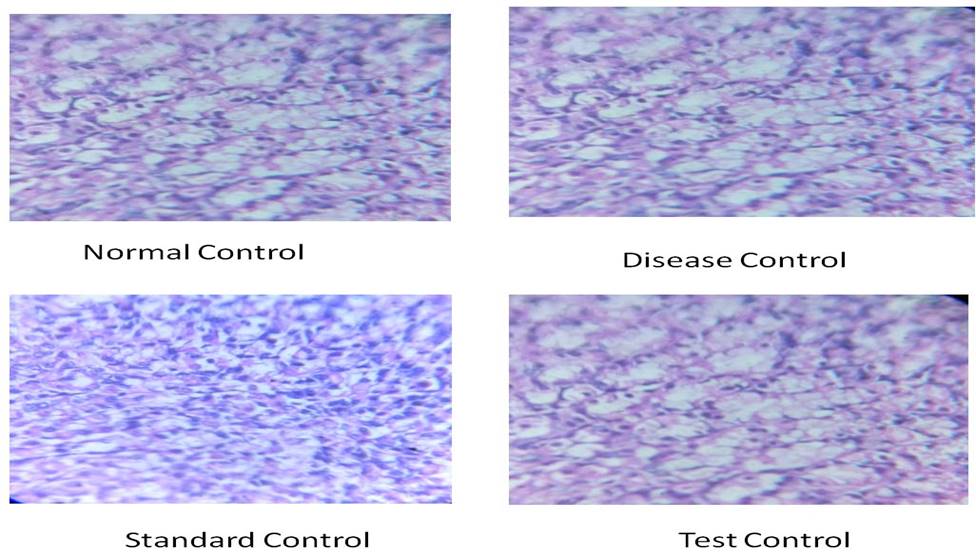
Figure 3. Histological study of stomach in diclofenac induced anti-ulcer
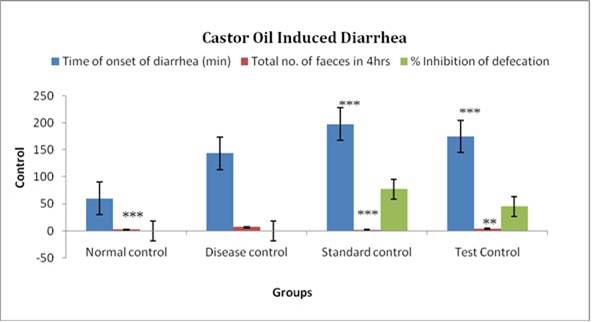
Figure 4. Results of different groups treatments when compared with control group in Castor oil induced diarrhea *P<0.05, **P<0.01, ***P<0.001.
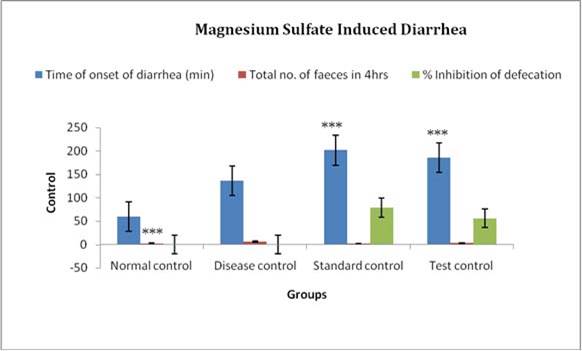
Figure 5. Results of different groups treatments when compared with control group in Magnesium sulfate induced diarrhea *P<0.05, **P<0.01, ***P<0.001.
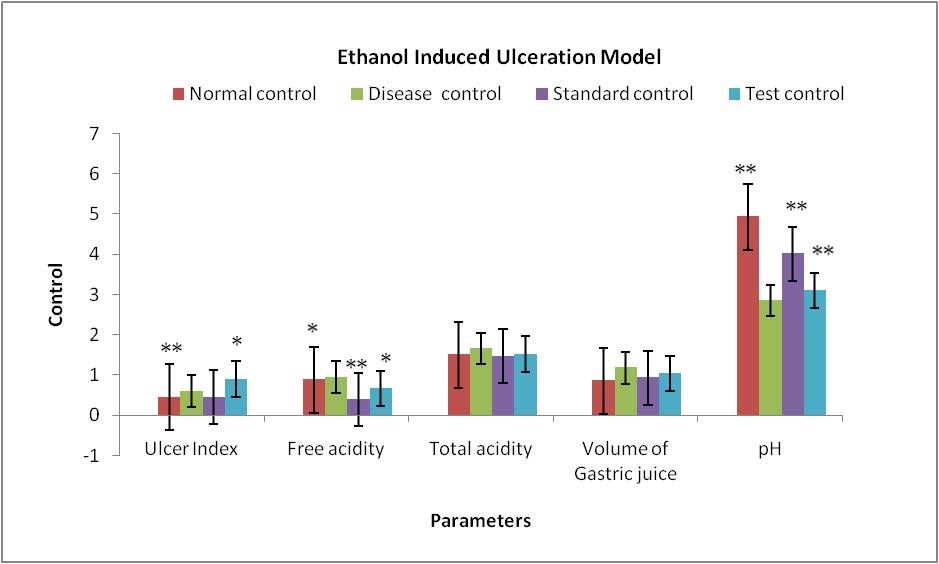
Figure 6. Results of different groups treatments when compared with control group in Ethanol Induced Ulceration Model *P<0.05, **P<0.01, ***P<0.001.
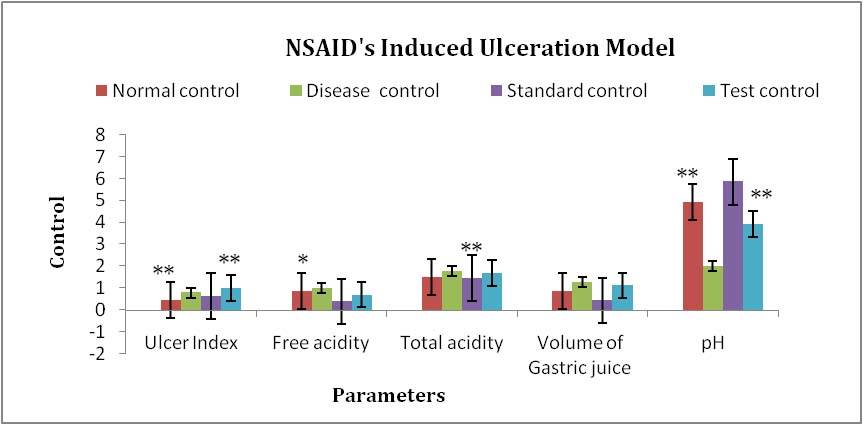
Figure 7. Results of different groups treatments when compared with control group in NSAID Induced Ulceration Model *P<0.05, **P<0.01, ***P<0.001.
Discussion
The present study was conducted to assess the anti-diarrheal and antiulcer properties of hydro-alcoholic extract of the aerial part of Tridax procumbens. Castor oil induced and magnesium sulfate induced diarrhea model for anti-diahorreal activity. Ethanol-induced and NSAIDs induced ulcer models for antiulcer activity were selected.
The hydro-alcoholic extract of Tridax procumbens contains phenols, tannins, flavonoids, saponins, terpenoids, glycosides, and anthraquinones as asserted by preliminary phytochemical screening tests, and most of these secondary metabolites were reported to have an antidiarrheal activity. Reports in the kinds of the literature showed that tannins have an antispasmodic and muscle relaxant effect, flavonoids inhibit prostaglandin E2-induced intestinal secretion, saponins inhibit histamine release, terpenoids inhibit the release of prostaglandins, and phenols reduce intestinal secretion and transit and have an astringent action. All these actions lead to the inhibition of diarrhea by decreasing intestinal secretion and motility. Therefore, the antidiarrheal activity of the plant extract may be produced by these chemical constituents.
In the preliminary phytochemical screening, the extract was positive for flavonoids, saponins, tannins, phenols, terpenoids, and alkaloids. Flavonoids are thought to increase mucosal prostaglandin content, decrease histamine secretion from mast cells by inhibition of histidine decarboxylase, inhibit Helicobacter pylori growth, act as free radical scavengers, and inhibit H+/K+-ATPase. Saponins may activate mucous membrane protective factors, and tannins render the outermost layer of the mucosa less permeable, for instance, to chemical irritation. Also, terpenoids and alkaloid compounds are also reported to have potent activity against gastric ulcers.
Diarrhea induced by castor oil and magnesium sulfate. Castor oil is a very effective laxative hydrolyzed into ricinoleic acid and stimulated fluid secretion, inhibited water and electrolyte absorption, reduced active Na+ and K+ absorption, and decreased Na+, K+-ATPase in the small intestine and colon. Castor oil also increased peristaltic activity and produced permeability changes in the intestinal mucosal membrane to electrolytes and water. Furthermore, ricinoleic acid can also lead to the release of endogenous prostaglandins, which play an important role in the modulation of GIT, stimulate motility and secretion, and cause diarrhea. In our study, the results showed that HATP could, in a dose-dependent manner, reduce castor oil-induced diarrhea as well as the number of diarrheal feces and a total weight of feces, which could be taken as antidiarrheal activities. Loperamide is used as a standard drug for diarrheal treatment. The therapeutic effect of Loperamide is due to its anti-motility and anti-secretory activity.
Magnesium sulfate has been reported to induce diarrhea by increasing the volume of intestinal content through the prevention of reabsorption of water. It has been demonstrated that it promotes the release of cholecystokinin from the duodenal mucosa, which increases the secretion and motility of small intestine and thereby prevents the reabsorption of sodium, chloride, and water.
Diclofenac, a known ulcerogenic especially in an empty stomach causes ulcers mostly on the glandular (mucosal) part of the stomach by inhibiting prostaglandin synthesis through the cyclooxygenase pathway. Prostaglandins function to protect the stomach from injury by stimulating the secretion of bicarbonate and mucus, maintaining mucosal blood flow and regulating mucosal turnover and repair. Suppression of prostaglandin synthesis by diclofenac results in increased susceptibility of the stomach to mucosal injury and gastro-duodenal ulceration. The extract was observed to significantly reduce mucosal damage in the NSAIDS induced ulcer model, suggesting the antiulcer effect of the extract.
In acute oral toxicity study, there were no behavioral changes seen up to 4 hrs and no mortality was observed up to the end of 24 hrs even at the maximum tested dose level of 2000 mg/kg per oral. The extract dose (300 mg/kg) was significantly reducing the diarrheal and ulcerative conditions in experimental rats. For the treatment of rats standard drug for diarrhea loperamide and ulcer ranitidine were used.
Conclusion
In conclusion, we can say that extract dose (300 mg/kg) of Tridax procumbens show significantly reducing the condition of diarrhea as well as an ulcer with the help of flavonoids, saponin, alkaloid, tannins, phenols and some other phytoconstituents. The plant extract was also found to have optimal safety margin based on the limit test at 2000 mg/kg dose level acute toxicity test. Therefore, the plant is potentially useful to develop plant-based products after further studies to identify the active principle and the mechanism of action.
Conflict of interest
None.
Acknowledgement
The authors are acknowledging to Ravishankar College of Pharmacy, Bhopal for Experimental Work and Dr. Zia Ul Hasan, Head of the Department Botany at the Safia College of Science, Bhopal (M.P.) for identification and authentication of the plant. Authors also want to thanks Dr. Kapil Khatri, Department of Pharmaceutics at Ravishankar College of Pharmacy, Bhopal (M.P.) for his valuable support.
References
Agrawal S, Mohale D, Talele GS. 2010. Pharmacological activities of Tridax procumbens (Asteraceae). Medicinal Plants 2(2):73-78.
Ashrafuzzaman MD, Raju GS, Muhammad Abeer IU, Saifuddin MS, Abu Zafor MS, Dipti D. 2016. Antidiarrheal activity of three medicinal plants in swiss albino mice. International Journal of Biomedicine 6(3):233-236.
CPCSEA Guideline for Laboratory Animal Facility.
Gerhard Vogel. 2008. Drug Discovery & Evaluation: Pharmacological Assays. Springer, 3rd edition second volume 1661-1717.
https://dailymed.nlm.nih.gov (Last accessed on 22 Sep 2018)
https://www.drugbank.ca (Last accessed on 22 Sep 2018)
https://www.healthline.com (Last accessed on 22 Sep 2018)
https://www.mayoclinic.org/ (Last accessed on 22 Sep 2018)
https://www.niddk.nih.gov (Last accessed on 22 Sep 2018)
Ingale AM, Pinnelli Venkata BK, Rajendran V. 2016. Experimental evaluation of the anti-ulcer activity of the ethanolic extract of grape (Vitis vinifera) seed in wistar albino rats against aspirin plus pylorus ligation induced gastric ulcer model. International Journal of Basic & Clinical Pharmacology 5(3):722-727.
Kaur M, Singh A, Kumar B. 2014. Comparative antidiarrheal and antiulcer effect of the aqueous and ethanolic stem bark extracts of Tinospora cordifolia in rats. Journal of Advanced Pharmaceutical Technology & Research 5(3):121-128.
Kethamakka SRP, Deogade MS, Jayanti V. 2014. (Tridax procumbens) - Unnoticed Medicinal plant by Ayurveda. Journal of Indian System of Medicine 2(1):1-20.
Khandelwal KR. 2003. Practical pharmacognosy techniques and experiments. 10th Edition Nirali Prakashan Pune 149-156.
Kokate CK. 1994. Practical pharmacognosy. 4th edition Vallabh Prakashan Delhi 107-111.
Mekonnen B, Asrie AB, Wubneh ZB. 2018. Antidiarrheal activity of 80% methanolic leaf extract of Justicia schimperiana. Hindawi Evidence-Based Complementary and Alternative Medicine 1-10.
Mir SA, Jan Z, Mir S, Dar AM, Chitale G. 2017. A concise review on biological activity of Tridax procumbens Linn. Organic Chemistry Current Research 6(177):1-4.
OECD 2001-guideline on acute oral toxicity (AOT), Environmental health and safety monograph series on testing and adjustment, No. 425.
Panneerselvam S, Arumugam G. 2011. A biochemical study on the gastroprotective effect of hydroalcoholic extract of Andrographis paniculata in rats. Indian Journal of Pharmacology 43(4):402-408.
Sathish R, Sahu A, Natarajan K. 2011. Antiulcer and antioxidant activity of ethanolic extract of Passiflora foetida L. Indian Journal of Pharmacology 43(3):336-339.
Seth Sweetser MD. 2012. Evaluating the patient with diarrhea: A case-based approach. Mayo clinic proceedings 87(6):596–602.
Tadesse WT, Hailu AE, Gurmu AE, Mechesso AF. 2014. Experimental assessment of anti-diarrheal and anti-secretory activity of 80% methanolic leaf extract of Zehneria scabra in mice. BMC Complementary and Alternative Medicine 14(460):2-8.
Tripathi KD. 2008. Essentials of Medical pharmacology. 6th edition JP brother’s medical publishers 613-614.
Vimala G, Shoba F, Gricilda. 2014. A review on antiulcer activity of few Indian medicinal plants. International Journal of Microbiology 1-14.
Wansi SL, Nguelefack-Mbuyo EP, Nchouwet ML, Miaffo D, Nyadjeu P, Wabo JP, Mbiantcha M, Keng- Efouet Pepin AN, Nguelefack TB, Kamanyi A. 2014. Antidiarrheal activity of aqueous extract of the stem bark of Sapium ellipticum (Euphorbiaceae).Tropical Journal of Pharmaceutical Research 13(6): 929-935.
www.drugs.com (Last accessed on 22 Sep 2018)