
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Bhagwati Sharan Sharma, N. S. Lodhi*, R. B. Goswami, Abhishek Banke
Sagar Institute of Research and Technology Pharmacy, Bhopal (M.P.) India
*Address for Corresponding Author
Dr. N. S. Lodhi
Department of Chemistry
Sagar institute of Research and Technology pharmacy, Bhopal (M.P.) India
Abstract
Objective: Tuberculosis (TB) Caused with poor patient compliance and intolerance at least partially due to adverse drug reactions (ADRs). A prospective observational and interventional healthcare teamwork study was carried out to implement a self-reporting pharmacovigilance system in TB patients through a knowledge-based approach in the civil Hospital at Narsinghgarh (M.P). Methods: A patient information form which was endorsed by the pulmonology associates was use as a tool for this study. A well trained and skilled clinical pharmacist educated the patients and enabled them to report the ADRs due to anti-tubercular drugs during counselling. Totally 100 patients enrolled in the study. 84 (84%) patients experienced 74 numbers of ADRs during the intensive phase therapy. Results: 100 patients recruited in our study, 84(84%) patients developed ADR’s. Most of ADR ‘s (26.19%) occurred in the age group of 14-25 years Males were more prone to develop ADR ‘s (59.52%) than females (40.48%). Nonsmokers (72.62%), non drinkers (76.19%), non-vegetarians (59.52%) and illiterate patients developed more ADR’s as compared to smokers (27.38%), drinkers (23.81%), vegetarians (40.47%) and literate (45.23%). Conclusion: Our study concludes that if a proper educational system is implemented, most of the patients were ready to report their ADR of any drug and thereby we can improve both patient adherence and reducing the severity of ADRs. It is suggested that the pharmacists should exhibit their vital role during TB therapy in TB centers, pulmonology departments and DOTS centers to guarantee a better patient care.
Keywords: Self reporting pharmacovigilance, Anti-TB drugs, Clinical pharmacist, ADR reporting system
Introduction
Tuberculosis (TB) is the most common communicable infectious disease on earth and remains out of control in many developing nations. Good patient adherence to the treatment regimens is the foundation stone to effective Anti-Tubercular Therapy (ATT) (Dipir et al., 2005). Alas, noncompliance is cited as the major problem to the control of tuberculosis at the level of public health and finally which escort to the drug resistance in case of TB (Erhabor et al., 2000).
ATT exhibits greater level of efficacy with a satisfactory degree of toxicity; however, combination treatment may produce severe adverse events. Important adverse effects are hepatitis, joint pain, skin rash, gastro intestinal upset (nausea/vomiting/GI upset), hyperuricemia, constipation, peripheral neuropathy, and visual disturbances (Mahmood et al., 2009). TB hampered with poor patient compliance and intolerance at least partially due to the ADRs. According to World Health Organization (WHO) and several other studies concluded that, the poor out-come was attributed to poor patient compliance, to primary multidrug resistance and to interruption partially due to ADR (WHO 1997) (Schaberg et al., 1996) and the towering incidence of TB infection has caused a high occurrence of morbidity and mortality which is partly due to serious ADRs induced by Anti- TB drugs (Begum et al., 2006).
Patient’s decisions to stop taking medications were influenced by a number of interacting factors (Munro SA et al., 2007). The lack of knowledge about the treatment and ATT induced ADRs are the two major factors which leads to the patient’s non-adherence to the TB therapy. A qualitative and quantitative study by Weiguo et al. ( Xu et al., 2009)stated that almost 16 factors which leads to the non adherence for the TB treatment. Majority of them are due to the lack of knowledge about the importance of the completion of therapy. Out of these 16 factors 37.80% (which is the highest percentage) of patients were non adherent due to the severe ADRs ( Xu et al., 2009). Schaberg et al. (Schaberg et al., 1996) were also stated that 26% of TB patients in the study population were discontinued therapy due side effects.
The influence of side effects – real, anticipated or interpreted on compliance to treatment was mentioned in a number of studies. Some patients reported stopping medication due to adverse effects while others complained that they were not educated about side effects and what to do to counter them (Munro et al., 2007). In-depth interviews among both TB patients and local doctors point out that ADR is a motive for treatment non-adherence. Worry of the risks of ADRs leads some TB patients to break off the treatment (Xu et al., 2009) “… I don’t want to take these pills, because they make me sick, they hurt me…” (Female TB patient, Bolivia) is an example for the above statement. Local health workers often cannot find out this discontinuation of treatment due to the lack of an ADR surveillance system under the current DOTS program (Xu W et al., 2009). Counselling of patients for timely hindrance, revealing and management of ADRs was also highly suggestive (Chhetri et al., 2008). Also it is already proved that patients were clearly willing to report symptoms which they believe to be due to a particular prescribed drug if they were informed about it. So it is fundamentally required a system for proper monitoring of ADRs due to anti tubercular drugs. Several studies were suggesting the significance of a new system for premature detection of ADR for a better patient care (Khalid et al., 2007).
This leads to taking a decision to do an intervention of a new health care teamwork approach with an intention to complete patient care during ATT with a special preference on ADR reporting system. The evidence of patient’s definite role in ADR and their willingness to report the ADR (Campbell et al., 1988) were planned to utilize in this study by expecting a good adherence. Here comes the importance of pharmaceutical care based approach to the TB patient. This study is one of the clinical pharmacy come health care team work oriented one, aiming for the best quality of life of tuberculosis patient during their therapy by implementing an educational approach to them regarding on both the therapy and how to counteract the possible ADRs during Anti Tubercular Therapy.
Anti-Tubercular Drugs: More than twenty drugs have been developed for the treatment of Tuberculosis. Most of them were developed some years ago. The drugs are used in differing combinations in different circumstances. For example, some Tuberculosis drugs are only used for the treatment of new patients who are very unlikely to have resistance to any of the Tuberculosis drugs. There are other drugs that are only used for the treatment of drug resistant Tuberculosis.
There are now starting to be some new Tuberculosis drugs, but there is not very much known about them, and they are still undergoing testing. The five basic or first line Tuberculosis drugs are
These are the Tuberculosis drugs that generally have the greatest activity against Tuberculosis bacteria. These drugs are particularly used for someone with active Tuberculosis disease who has not had Tuberculosis drug treatment before. All the other Tuberculosis drugs are generally referred to as second line or reserve Tuberculosis drugs. All the drug names have abbreviations which are either one, two or three letters. There are also trade or brand names, which is the name by which a drug is known when it is being sold in a particular country and is made by a particular manufacturer. For example, ethambutol is known in India by a variety of trade names which include Abietol (made by Alpic Remedies), Actuate (made by Biocon Genetics) and Albutol (Alkem Laboratories).
In other countries the trade name will be different. For example, ethambutol is sometimes referred to as Myambutol. Isoniazid is sometimes called AKT4 (Lupin Laboratories).
Tuberculosis drug treatment for new patients
Patients who have not had any Tuberculosis treatment before, or they have had less than one month of anti-Tuberculosis drugs, are considered to be new patients. New patients are presumed to have drug susceptible Tuberculosis (Tuberculosis which is not resistant to any of the drugs) unless there is a high level of isoniazid resistance in new patients in the area. The other people who may have drug resistant Tuberculosis are people who have developed active Tuberculosis disease after they have been in contact with a patient who is known to have drug resistant Tuberculosis. For new patients the World Health Organization (WHO) recommends that they should have six months of Tuberculosis drug treatment. This should consist of a two month
intensive treatment phase followed by a four month continuation phase. For the two months
intensive Tuberculosis drug treatment phase they should receive:
With rifampicin (R/Rif) for the continuation Tuberculosis drug treatment phase. It is essential to take several Tuberculosis drugs together. If only one Tuberculosis drug is taken on its own, then the patient will very quickly become resistant to that drug. It is recommended that patients take the Tuberculosis drugs every day for the six months, although taking them three times a week is possible in some circumstances. It is extremely important that all the recommended Tuberculosis drugs are taken for the entire time. The amount of any drug that a patient needs to take depends on the patient‘s weight. If only one or two of the Tuberculosis drugs are taken, or the treatment is interrupted or stopped early, then the treatment probably won ‘t work. This is because the Tuberculosis bacteria that a patient has, develops resistance to the Tuberculosis drugs. Not only is the patient then still ill, but to be cured they then have to take drugs for the treatment of drug resistant Tuberculosis.
Tuberculosis drugs for the treatment of drug resistant Tuberculosis
Recommendations before May 2016
Before May 2016 the Tuberculosis drugs which were used as treatment for drug resistant Tuberculosis were those listed below. All the Tuberculosis drugs in Group or class 1 are first line drugs. Another first line drug is streptomycin which is with the other injectable agents in Group 2. All the drugs in Groups 2 to 5, apart from streptomycin, were referred to as second line or reserve Tuberculosis drugs 6. The first four groups of Tuberculosis drugs listed below were those that were mainly used for the treatment of drug resistant Tuberculosis. The fifth group of Tuberculosis drugs were some drugs that were unknown in how effective they were in the treatment of Tuberculosis. They could however be tried when there was no other option. They were sometimes used in the treatment of totally drug resistant Tuberculosis.
Change of recommendations in May 2016: In May 2016 the WHO changed their recommendations on the use of drugs for the treatment of drug resistant Tuberculosis 7.This was partly because of the Bangladesh regimen, which appeared to show that a shorter regimen could be just as successful as a longer regimen. 8 There had also been a number of other studies which had shown a similar result. There had however been no randomized control trial, but the WHO had decided that the treatment of MDR-Tuberculosis was too important for there not to be new recommendations. This regrouping is intended to guide the design of conventional regimens. For shorter regimens lasting 9-12 months the composition is usually standardized.
The Primary objective of this study was to find out the commonly encountered ADRs among patients on anti-tubercular therapy (ATT) in the Civil hospital, Narsinghgarh (M.P.) and assessment of their severity and causality. The aim of this study was also to find the missing links (if any) and suggest a more reliable, precise and accurate Pharmacovigilance technique for Antitubercular drugs used in a Community Healthcare Hospital. The purpose was also to find out and highlight if the significant ADRs are not being reported due to loop hole in the process.
Material and methods: The present study was carried out in the in-patient and out- patients Medicine Department of Civil Hospital, Narsinghgarh (M.P.) after obtaining the necessary permission from the Block Medical Officer, Dr. Gaurav Tripathi. Informed consent was obtained from the conscious patients or from their relatives if the patients were unconscious. The study was conducted for a period of one year.
Inclusion Criteria
All in-patients admitted and out- patients due to Tuberculosis in Civil Hospital, Narsinghgarh (M.P.) and who were willing to participate in the study.
Exclusion Criteria
Patients who were not willing to participate in the study. Source of Data Data was collected from case sheets, lab reports and prescriptions of Tuberculosis patients admitted in Civil Hospital, Narsinghgarh (M.P). The data was collected by making ward rounds, identifying the Tuberculosis cases and collected the data. A visit to hospital was given to check for any new cases. Details of each Tuberculosis case was recorded in the data collection form which included details of age, gender, region, literacy levels, occupation, marital status, habits, past history, month & time of exposure to infection, signs & symptoms, treatment given, current treatment including DOTS, lab reports, length of hospital stay and patient status after treatment. It was a prospective observational study which was conducted in the Antituberculosis department of Govt. Civil Hospital, Narsinghgarh, District Rajgarh (M.P) of India which is a 30 bedded Government hospital. For Recording of Data following methods were used:
Online database for reporting Our study was open, prospective, observational, non-comparative study which was conducted in the Civil Hospital, Narsinghgarh (MP). One hundred sputum positive patients, of pulmonary Tuberculosis on DOTS category I, of either sex, in the age group of 14 years to 65 years, from out-patient department and patients admitted in wards of the Department of Tuberculosis and Chest diseases Hospital from January 2018 to December 2018 were recruited. The approval of Block Medical Officer, Shri Gaurav Tripathi was taken before the start of study. Written informed consent was taken from patients in their vernacular language. The study subjects on DOTS Cat I were put on Short Course Chemotherapy Regimen comprising of 600mg Isoniazid(H), 450mg Rifampicin(R), 1200mg Ethambutol(E) and 1500mg Pyrazinamide(Z). The patients were then monitored for adverse drug reactions for 60 days. Proforma was designed to record detailed history, examinations, laboratory investigations, and ADR ‘s of study subjects. Follow up was done at 30 and 60 days during which patients were actively enquired of any adverse drug reactions and the same were recorded on proforma. Physical characteristics which include body weight, height, BMI, BP and laboratory investigations like CBC, SGOT, SGPT, urea, creatinine, uric acid, sputum microscopy and serum bilirubin were done at 0, 30 and 60 days. Causality assessment was done by using WHO-UMC causality criteria and severity assessment was done by using Hartwig ‘s severity scale. Demographic characteristics of patients, incidence of various ADR ‘s and their impact on sputum conversion rate were assessed and data was analyzed using Chi-square test (x2) and ANNOVA (F test) test.
Results and Discussion
For the period 1st quarter to 4th quarter 2018 (annually) following number of patients were identified with Tuberculosis at Civil Hospital, Narsinghgarh (M.P.) as shown in table 1.
Table 1. number of patients were identified with Tuberculosis at Civil Hospital, Narsinghgarh (M.P.)
|
Territory |
Year |
NSP |
NSS |
NEP |
Relapse |
Failure |
TAD |
Others |
Total |
|
Narsinghgarh |
2018 |
182 |
280 |
80 |
0 |
0 |
0 |
111 |
653 |
NSP: New spectrum Positive
NSN: New Sputum Positive
NEP: New Extra Pulmonary
Relapse: Patients, who have developed Tuberculosis again even after successful eradication in first instance.
Failure: Patients, which do not respond to the medication throughout the treatment cycle and remains Sputum Positive.
TAD: Treatment After Default, Patients which stops taking medication for more than 1 month during the treatment cycle will fall under this category if they come back for treatment again.
Others: All other patients with Tuberculosis will fall under this category, where the sufficient historical data is not available for them. Out of 100 patients recruited in our study, 84(84%) patients developed ADR ‘s. Most of ADR ‘s (26.19%) occurred in the age group of 14-25 years Males were more prone to develop ADR ‘s (59.52%) than females (40.48%). Nonsmokers (72.62%), non drinkers (76.19%), non vegetarians (59.52%) and illiterate patients developed more ADR ‘s as compared to smokers (27.38%), drinkers (23.81%), vegetarians (40.47%) and literate (45.23%) as shown in Table 2.
Table 2. Baseline characteristics of the patients studied
|
Parameters |
Developed ADR’s |
Not developed ADR’s |
x2 value |
p-value |
|
|
Age (years) |
14-25 |
22 (26.19%) |
2 (12.50%) |
11.971 |
0.018 |
|
26-35 |
15 (17.85%) |
1 (6.25%) |
|||
|
36-45 |
15 (17.85%) |
4 (25%) |
|||
|
46-55 |
13 (15.47%) |
8 (50%) |
|||
|
56-65 |
19 (22.61%) |
1(6.25%) |
|||
|
Sex |
Male |
50 (59.52%) |
10 (62.5%) |
0.003 |
0.956 |
|
Female |
34 (40.48%) |
6 (37.5%) |
|||
|
Smoking status |
Non smokers |
61 (72.62%) |
12 (75%) |
0.012 |
0.912 |
|
Smokers |
23 (27.38%) |
4 (25%) |
|||
|
Drinking status |
Non drinkers |
64 (76.19%) |
13 (81.25%) |
0.019 |
0.907 |
|
Drinker |
20 (23.81%) |
3 (18.75%) |
|||
|
Dietary pattern |
Vegetarians |
34 (40.47%) |
6 (37.5%) |
0.003 |
0.956 |
|
Non-vegetarians |
50 (59.52%) |
10 (62.5%) |
|||
|
Education status |
Literate |
38 (45.23%) |
5 (31.25%) |
0.578 |
0.447 |
|
Illiterate |
46 (54.76%) |
11 (68.75%) |
|||
The most common ADR in our study was GI upset (38.13%) and hepatitis (35.59%) Frequencies of other ADR‘s were as shown in Figure 1
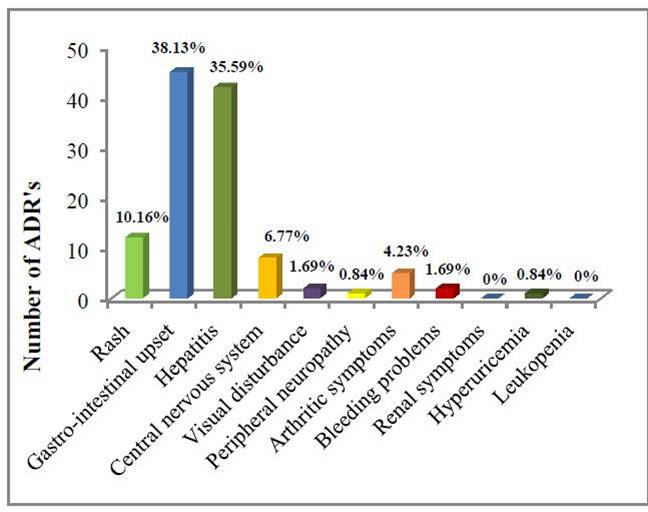
Figure 1. Incidence of various ADR‘s
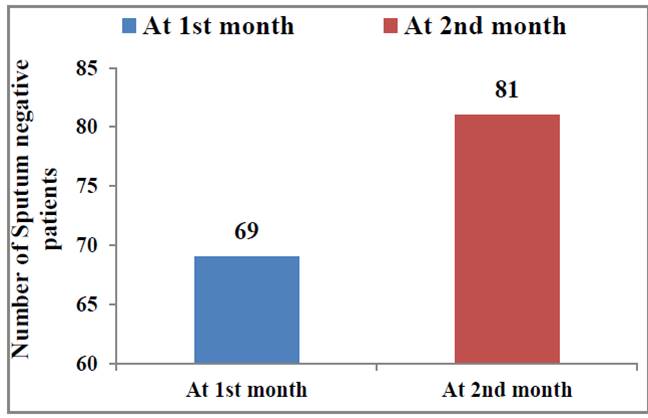
Figure 2. Number of patients converted to sputum negative at the end of 1st and 2nd month
Sputum Conversion Rate (SCR) at the end of two months of treatment in the patients with ADR‘s and without ADR‘s was 79.77% and 87.5% respectively as shown in Figure.
Relationship of occurrence of ADR’s with duration of therapy.
Table 3 demonstrates that 59 (50%) of ADR ‘s occurred within 1-15 days after starting therapy, followed by 41 (34.74%), 11 (9.32%) and seven (5.93%) within 16-30 days, days and 40-60 days, respectively.
Table 3. Relationship of occurrence of ADR’s with duration of therapy
|
No. of days of starting therapy |
No. of ADR’s |
Percentage |
|
1-15 |
59 |
50 |
|
16-30 |
41 |
34.74 |
|
31-45 |
11 |
9.32 |
|
46-60 |
7 |
5.93 |
Out of 118 ADR ‘s, 42 (35.59%) were of drug induced hepatitis. There was a rise in SGOT, SGPT and bilirubin levels at 30 days of treatment, followed by a fall in levels of these laboratory parameters at 60 days as shown in table. The change in SGOT, SGPT and bilirubin levels at 0, 30 and 60 days was statistically significant with p- value of 0.001, 0.011 and 0.013, respectively. Mean value of hemoglobin, BMI and body weight increased with duration of treatment but the value was not statistically significant. Causality assessment of various ADR ‘s as per WHO-UMC criteria reveals that majority of cases 60 (71.43%) belonged to probable, followed by 20 (23.81%) cases of possible and four (4.76%) cases of certain as shown in Figure 3.
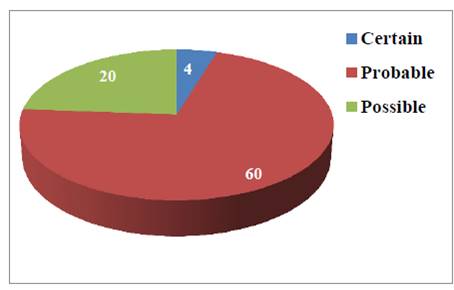
Figure 3. Causality of ADR’s as per WHO-UMC causality criteria
Causality of ADR’s as per WHO-UMC causality criteria. Mean change in various parameters at 0, 30 and 60 days. Table 4.
Table 4. Mean change in various parameters at 0, 30 and 60 days
|
Parameters |
Day 0 |
Day 30 |
Day 60 |
F test |
p- value |
|
Body weight |
45.84±10.77 |
46.97±10.73 |
47.95±10.57 |
0.975 |
0.378 |
|
BMI |
18.13±3.94 |
18.58±3.93 |
19.01±3.96 |
1.224 |
0.296 |
|
Hemoglobin |
9.24±1.81 |
9.37±1.49 |
10.33±2.88 |
1.332 |
0.266 |
|
WBC |
10525.12±2498.12 |
9072.3±1923.01 |
8411±1302.05 |
34.093 |
0.001 |
|
SGOT |
41.13±12.85 |
53.24±26.40 |
45.87±18.91 |
9.159 |
0.001 |
|
SGPT |
43.98±14.90 |
52.1±23.91 |
46.84±18.06 |
4.543 |
0.011 |
|
Bilirubin |
0.72±0.45 |
0.92±0.90 |
0.70±0.20 |
4.417 |
0.013 |
From the Figure 3, showing analysis of severity assessment of ADR‘s on Hartwig‘s scale, it is evident that most of the cases (55) were of level 4 ( moderate) severities where the treatment with suspected drug was withheld and length of stay was more than one day.
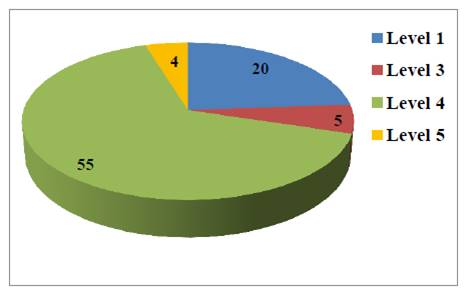
Figure 4. Categories of ADR‘s as per Hartwig’s severity scale
Concurrent medications prescribed for Treatment
|
Concurrent Medication |
Number of Patients out of 100 |
|
Analgesics |
27 |
|
Antibiotics |
100 |
|
Antiemetics |
34 |
|
Antiulcers |
7 |
|
CNS depressants |
11 |
|
Diuretics |
0 |
|
Nutritional supplements |
91 |
|
Anti-Histaminic |
34 |
|
Supportive therapy |
16 |
The duration of stay in the hospital varied from seven to twenty days depending on severity of infection. Hospitalization days ranged from 4 to 20 days with a mean of 10-15 days. Out of 88 cases, 33 patients recovered partially and 11 patients relapsed with the positive sign of tuberculosis.
Anti-Tuberculosis drugs could cause significant adverse effects both in magnitude and severity. Results of our study reveal that 84% of patients developed one or more ADR‘s and a total of 118 ADR‘s occurred in our study.
This relatively high incidence is in concordance with the study done by Koju et al and the study done by Ramanand et al which showed the incidence of 80% and 92.5% respectively.10,11 Most common ADR was GI upset 45 (38.13%), followed by hepatitis 42 (35.59%), rash 12 (10.16%), CNS 8 (6.77%), arthritic symptoms 5 (4.23%), visual disturbance 2 (1.69%), bleeding problems 2 (1.69%), hyperuricemia 1 (0.84%) and peripheral neuropathy 1 (0.84%). Various other studies like Sahithi et al, Honnaddi et al and Sinha et al also showed higher incidence of gastro-intestinal problems.12-14 Out of total 118 ADR‘s, 59 (50%) ADR‘s occur within 1-15 days after starting therapy, followed by 41 (34.74%), 11 (9.32%) and seven (5.93%) within 16-30 days, 31-45 days and 40-60 days, respectively. This is in concordance with the study done by Kheirollah et al and Abideen et al in which maximum number of ADR‘s were seen within lag period of 1-15 days. Causality assessment revealed that most of ADR‘s were in probable category and severity assessment revealed that most of ADR‘s belong to level 4 (Moderate severity).
Discussion
The study finding of occurrence of majority of ADR‘s within first 30 days of treatment calls for intensive training of health personnel in identification and timely management of ADR‘s during this crucial period. There is a need of incorporating pharmacovigilance programme into this vital health programme for more comprehensive monitoring of Tuberculosis patients on DOTS. Counselling of patients for timely prevention, detection, and management of ADR‘s will build confidence of patients, decrease non-adherence and dropouts, and thus result in better treatment outcomes. This emphasizes the importance of developing strategies at the programme level to deal with ADR‘s both to improve the quality of patients care and to control Tuberculosis safely. Tuberculosis is a major problem in urban as well as ruler setting leading to hospitalization and even death of individuals.
Having information about the commonly drugs used for Tuberculosis and updated guidelines for the management of the same would be helpful to the clinicians as a quick reference before treating the patients. The PV in India has become an important public health issue as regulators, drug manufacturers, consumers, and healthcare professionals are faced with a number of challenges. The PV in India continues to grow, evolve, and improve. India is the largest producer of pharmaceuticals and now emerging as an important clinical trial hub in the world. Apparently, the requirements for professional specialization, a combined view on PGx and clinical requirements are needed. That helps to identify factors that increase the risk of unwanted outcomes from drug therapy and prior to commencing drug treatment and in tailoring drug treatment for individual patients. The PV has also involved in Data Mining Technology in spontaneous reports submit to the national surveillance systems. The PVPI is coordinated at IPC through NCC under the control of Indian Government to generate an independent data on safety of medicines, which will be at par with global drug safety monitoring standards. National and regional PV systems are well-adapted bodies, attuned to the intricate collection and analysis of ADR data that leads to timely alerts and interventions to protect population health. Furthermore, it is responsible in India of entire campaign to improve PV knowledge and increase the number of ADRs reports up to the gold standard level established by the WHO. The adverse events reported by PV system will potentially benefit to the community due to their proximity to both the population and public health practitioners, in terms of language and knowledge of the lifestyle and habits of patients, enables easy contact with reporters, for example by telephone, Email, text massages by mobile phones. The development of new and effective medicinal products makes a positive contribution to the health and well being of individuals. However, there is a need to improve PV systems to more effectively monitor and take action on safety issues associated with medicines to enhance their contribution to public health. Hence, PV for medicinal product safety to help the patients get well and to manage optimally or ideally, avoid illness is a collective responsibility of industry, drug regulators and clinicians and other healthcare professionals. The financial support and future projects should help to achieve a more comprehensive PV activity in India.
Future Prospects: As future prospects increase, PV systems capable to detect new ADRs, and taking regulatory actions are needed to protect public health. Little emphasis has been put into generating information that can assist a healthcare professional or a patient in the decision-making process. The gathering and communication of this information is an important goal of PV. Information about the safety of drug active surveillance is necessary. When develop new methods for active post-marketing surveillance, one has to keep in mind that the important to collect complete and accurate data on every serious reported event. Spontaneous reporting is a useful tool in generating signals, but the relatively low number of reports received for a specific association makes it less useful in identifying patient characteristics and risk factors. PV methods must also be able to describe which patients are at risk of developing an ADR. As a source of information, the PV approach would be consistent with the growing patient involvement in drug safety. The PG could play a role in identifying individual risk factors for the occurrence of certain ADRs. In the future, PV has to concentrate on the patients as a source of information in addition to the more traditional groups, such as the health professionals. At present, the DCGI should act quickly to improve PV so as to integrate Good Pharmacovigilance Practice (GPP) into the processes and procedures to help ensure regulatory compliance and enhance clinical trial safety and post marketing surveillance. An appropriately working PV system is essential if medicines are to be used carefully. It will benefit healthcare professionals, regulatory authorities, pharmaceutical companies and the consumers. It helps pharmaceutical companies to monitor their medicines for risk. Post-marketing PV is currently a challenging and laborious process, not only industry-wide, but also for regulatory agencies.
Conclusion: The aim of the PV is to receive the information, documentation of the work and knowledge online while giving priority to the new and important safety issues. Non serious events have less priority than serious events but important in comparing the changes in health, although they are also screened routinely. The number of Adverse Drug Reactions (ADRs) reported resulted in an increase in the volume of data handled, and to understand the pharmacovigilance, a high level of expertise is required to rapidly detect drug risks as well as to defend the product against an inappropriate removal. Recently, pharmacovigilance has been confined, mainly to detect adverse drug events that were previously either unknown or poorly understood. Pharmacovigilance is an important and integral part of clinical research and these days it is growing in many countries. Today many pharmacovigilance centers are working for drug safety monitoring in this global pitch, however, presently pharmacovigilance faces major challenges in aspect of better safety and monitoring of drugs.
Acknowledgement
I would like to thanks my esteemed guide Professor Dr. N.S. Lodhi & my co-supervisor Ms. Abhishek Banke, for their valuable guidance and inspiration toward this innovative work, which made this project work possible. I would like to thanks Mrs. Suchi Thakur for her continuous support and understanding nature towards this project work. I would like to thanks Dr. Gaurav Tripathi (BMO, Narsinghgarh), Dr. Surendra Mittal (DTO, Rajgarh), Mr. Narendra Bansal, Mr, Mahendra Pahadi, and Mr. Prahlad Singh Ucharia from Civil Hospital Narsinghgarh for permitting me and providing necessary assistance and continuous support during this project work.
References
Alagiriswami B, Ramesh M, Parthasarathi G, Basavanagowdappa H. 2009. A study of clinical pharmacist initiated changes in drug therapy in a teaching hospital. Indian Journal of Pharmacy Practice, 2(1).
Bello SI, Itiola OA. 2010. Drug adherence amongst tuberculosis patients in the University of Ilorin Teaching Hospital, Ilorin, Nigeria. African journal of pharmacy and pharmacology, 4(3):109-14.
Campbell JP, Howie JG. 1988. Involving the patient in reporting adverse drug reactions. The Journal of the Royal College of General Practitioners, 38(313):370-1.
Chhetri AK, Saha A, Verma SC, Palaian S, Mishra P, Shankar PR. 2008. A study of adverse drug reactions caused by first line anti-tubercular drugs used in Directly Observed Treatment, Short course (DOTS) therapy in western Nepal, Pokhara. JPMA, 58(531).
Dipiro JT, Talbert RL, Yee GC, Matzke GR, Barbara GW. Tuberculosis: Pharmacotherapy A Pathophysiologic Approach, (6thedn).
Erhabor GE, Aghanwa HS, Yusuph M, Adebayo RA, Arogundade FA, Omidiora A. 2000. Factors influencing compliance in patients with tuberculosis on directly observed therapy at Ile-Ife, Nigeria. East African medical journal, 77(5).
Government of India 2006. TB India 2006, RNTCP Status Report, DOTS for All-All for DOTS, Ministry of Health and Family Welfare, New Delhi.
Jarernsiripornkul N, Chaisrisawadsuk S, Chaiyakum A, Krska J. 2009. Patient self-reporting of potential adverse drug reactions to non-steroidal anti-inflammatory drugs in Thailand. Pharmacy world & science, 31(5):559-64.
Jarernsiripornkul N, Krska J, Capps PA, Richards RM, Lee A. 2002. Patient reporting of potential adverse drug reactions: a methodological study. British journal of clinical pharmacology, 53(3):318-25.
Johnson J, Kagal A, Bharadwaj R. 2003. Factors Associated with Drug Resistance in Pulmonary. Indian Journal of Chest Diseases and Allied Sciences, 45:105-9.
Khan HU, Khan MH, Khan AM, Noor-ul-Iman KH, Habib H. 2009. Anti-tuberculous therapy induced liver Injury: frequency, management and outcome. Journal of Medical Sciences, 17:99-102.
Khan MA, Walley JD, Witter SN, Shah SK, Javeed S. 2005. Tuberculosis patient adherence to direct observation: results of a social study in Pakistan. Health policy and planning, 20(6):354-65.
Kochi A. 1991. The global tuberculosis situation and the new control strategy of the World Health Organization. Tubercle, 72(1):1-6.
Mahmood K, Hussain A, Jairamani KL, Talib A, Abbasi BU, Salkeen S. 2007. Hepatotoxicity with antituberculosis drugs: the risk factors. Pakistan journal of medical sciences, 23(1):33.
Nahar BL, Hossain AM, Islam MM, Saha DR. 2006. A comparative study on the adverse effects of two antituberculosis drugs regimen in initial two-month treatment period. Bangladesh Journal of Pharmacology, 1(2):51-7.