
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Thamizh Selvam N.1, Vasanth Kumar K.G.2, Acharya MV.3
1Assistant Director -Scientist-II (Biochemistry),
Central Ayurveda Research Institute- Neuromuscular and Musculoskeletal Disorders,
(Central Council for Research in Ayurvedic Sciences, Ministry of AYUSH, Government of India, New Delhi.)Thrissur, Kerala- 679 531. India.
2Assistant Director-Scientist III (Chemistry), Central Ayurveda Research Institute- Neuromuscular and Musculoskeletal Disorders,
(Central Council for Research in Ayurvedic Sciences, Ministry of AYUSH, Government of India, New Delhi.)Thrissur, Kerala- 679 531. India.
3Director, CARI-N&MSD,
Central Ayurveda Research Institute- Neuromuscular and Musculoskeletal Disorders,
(Central Council for Research in Ayurvedic Sciences,Ministry of AYUSH, Government of India, New Delhi.)Thrissur, Kerala- 679 531. India.
Address for Corresponding Author:
Dr. N. ThamizhSelvam Ph.D.,
Assistant Director -Scientist-II (Biochemistry)
Central Ayurveda Research Institute- N&MSD
Cheruthuruthy, Thrissur, Kerala- 679 531. India.
Abstracts
Objective: Ipomoea pes-tigridis L. is a flowering plant of the Convolvulaceae family. This family comprises the plants having high industrial, pharmaceutical, scientific and cultural significance. Material and methods: The present study was performed to evaluate the physico chemical, phytochemical, spectroscopical characteristics of the plant. The aqueous, ethanol and petroleum ether extracts of Leaf and Stem of I. pes-tigridis was prepared and analyzed. Results: Physico- chemical analysis showed the details of extractive portion of the plant and ash composition. Phytochemical Analysis revealed the presence of Tannins, Terpenoids, Glycosides and alkaloids in leaf and stem at varying concentrations. The spectroscopic studies revealed the pattern of major compounds present in the leaf and stem of I. pes-tigridis.
Keywords: Ipomoeapes-tigridis, spectroscopy, Tiger Foot Morning Glory, Phytochemical studies
Introduction
The herbal medicines occupy distinct position right from the olden days to present day. Medicinal herbs have been used as traditional primary healthcare agents, especially in Asian countries. The use of plants as a therapeutic material due to their chemical substances of medicinal value is very common in all over the world from ancient time (Prabhu et al 2011; Thamizh Selvam et al 2015; Ali et al 2012). Ipomoea pes-tigirids L. is one of the ethno-medicinal plants of India that has much folk lore. The plant has been studied for its various biological activities such as antimicrobial properties, cytotoxic property, laxative, analgesic and curative studies (Ramesh, 2010; Chodhury et al 2014; Begum et al 2015; Meira et al 2012; Silva, 2002). In India, the plant is widely used for wound healing. The tribal community in Kerala state of India use the herb for various painful conditions like headaches, swellings, poisonous stings, snake bites etc (Pawar and Patil, 2004; Bhellum, 2012; Danial, 1975; Diego and Harbornea, 1993).
Even though the plant is claimed for various activities, the literature survey revealed that there was not much research studies carried out in the aspects of detailed chemical characteristics and compound profiles of the plant parts. So, the present study has been taken up to evaluate Physico chemical, Phytochemical, and Spectroscopy characteristics of Leaf and Stem of I. pes-tigridis.
Ipomoea pes-tigridisL. is a twining, herbaceous, hairy, annual vine. This plant belongs to the family Convolvulaceae and is commonly known as “Tiger Foot Morning Glory” in English and locally known as ‘Pulichuvadi’ or ‘Pulichuvadu’ in Malayalam (Sahu and Gupta, 2013; Pawar and Patil, 2004).
It is usually found in bush land, riverside, cultivated ground and sandy soil. All parts of the plant covered with long, spreading, pale or brownish hairs. The leaves are rounded, 6-10 cm in diameter, palmately 5-9 lobed, heart-shaped at the based and hairy on both surfaces. The lobes of the blade are elliptic, with narrowed base and rounded sinuses (Von Rheede, 2003; Chopra et al 1995; Neeli and Dixit, 2007). The 5- lobed leaf resembles tiger’s paw, which inspired its common name, as well as the botanical species name pes-tigridis. The flowers occur in axillary, head, usually only one opening at a time. The sepal is green colour and 1 cm long; flowers are white and about 4 cm long. Flowers open at evening time and fade next morning.
Taxonomical details (Plant Database 1996; Plant Database 2000)
Botanical Name : Ipomoea pes-tigridisL.
Kingdom : Plantae
Family : Convolvulaceae
Genus : Ipomoea
Species : Ipomoea pes-tigridis L.
Synonyms : Ipomoea bilobaForsk., Convolvuluspes-caprae L.
Vernacular Names : Panchpatia (Hindi), Tiger’s Foot (English), Pulichuvadi (Malayalam)
Materials and Methods
Plant collection and Authentication
The plant materials were collected from Western ghat region of Kerala. The plant was authenticated by Dr. N. Sasidharan, Taxonomist, Kerala Forest Research Institute, Thrissur. The Specimen voucher is maintained in the Department of Biochemistry, NRIP.
Chemicals and Reagents
Chemicals and Regents of AR grade purchased from Spectrum India Ltd, Merck India Ltd, Nice India Ltd, were used.
Extraction
The plant extraction was carried out using aqueous, ethanol and petroleum ether as solvents. The shade dried plant materials such as leaf and stem were individually extracted.
Phytochemical Analysis
The phytochemical analysis of the study material was carried out as per the standard protocols. It comprised of various tests including Salkowski test, Dragendorff’s test, Keller Killani test and Ellagic acid test protocols ( Reddy and Mishra, 2012; Kavit et al 2013; Vinoth and Manivasagaperumal, 2013; Tiwari et al 2011; Sofowara, 1982; Moses et al 2014; (21-27).
Physico-chemical Analysis
Physico-chemical analysis such as moisture content, total ash, water soluble ash, acid insoluble ash, water soluble extractive and alcohol soluble extractive were carried out as per the standard protocol mentioned below (Suhas and Vilas, 2014; Pratap et al 2012; Rao et al 2014; Soni et al 2011; Lohar, 2007).
Loss on drying
About 4g drug after accurately weighing was taken in a tared evaporating dish. The sample was placed in Hot air oven at 1050C for 5hrs and weighed. The process was continued i.e. the drying and weighing at one hour interval until difference between two successive weighing correspond to not more than 0.25%. The constant weight is noted when two consecutive weighing after drying for 30min and cooling for 30min in a desiccators shown not more than 0.01g difference.
Total ash
The 2-3g of the air dried drug is incinerated in a silica crucible at temperature not exceeding 4500C until free from carbon, cool and weigh. The % of ash was calculated with reference to air dried drug.
Water soluble ash
The ash obtained from Total ash procedure was boiled with 25ml water and insoluble matter was collected in a Gooch crucible on ashless filter paper. Then, it was washed with hot water and ignited for 15min at a temperature not exceeding 4500C. Subtract weight of insoluble matter from weight of ash, the difference in weight represents Water soluble ash with reference to air dried drug.
Acid Insoluble ash
The ash was boiled for 5min with 25ml dil. HCl and mix with glass rod. Filtered with Whatman paper No.1. to remove the acid and then add distilled water. For checking the acid content in the filterate, 1 drop of methyl orange was added. Appearance of yellow colour confirmed that there is no acid content. Then, Take the filter paper and fold it. Put in the crucible and incinerate up to 500C until filter paper burns and 1000C up to white colour.
Water soluble extractive
Accurately weighed sample (1g) taken in a Iodine flask and 20ml Chloroform-water mixture (0.25:100) added to the flask and kept closed for 24hrs. The next day solution was filtered and from the filtrate about 5ml was transferred to previously weighed evaporating dish and evaporated to dryness. After drying weight was noted and calculated the extractive value.
Alcohol soluble extractive
Macerate 5g (1g) of air dried drug coarsely powdered with 100ml (20ml) of alcohol of specified strength in a closed flask for 24hrs, shaking frequently during 6 hours and allowing to stand for 18 hours. Filter rapidly taking precautions against loss of solvent, evaporate 25ml of filtrate to dryness in a tared flat bottom shallow dish and dry at 1050C to constant weight and weigh. Calculate % of Alcohol soluble extractive with reference to air dried drug.
Spectrophotometric Analysis
Spectrophotometric characteristics were analyzed for understanding the basic chemical profiling of Ipomoea pes-tigridis and to compare the proposition of major compounds among different parts of the plant. The ethanolic extract of different parts such as leaf and stem of I. pes-tigridiswas taken at three different concentrations i.e. 1/10, 1/20, 1/40, 1/160. The extract was dissolved in ethanol for spectroscopic analysis, and pure ethanol was used as blank. The spectrum characteristics were measured after standardizing the procedures and analysis was confirmed by carrying out triplicate analysis at 200-800 nm (Anand and Gokulakrishnan, 2012; Santhanakrishnan et al 2014; Janakiraman et al 2011; Sathish et al 2012; Harborne, 1998).
Result and Discussion
The Physico-chemical analysis Ipomoea pes-tigridis showed the content of ash as 10.03% and 12.21 % for stem and leaf respectively. The acid insoluble ash and water soluble ash of leaf is found to be 0.63% and 4.80% respectively and it was 0.04 and 7.70 % for stem. The alcohol soluble extractive percentage was higher in leaf (6.52%) comparing with stem (4.57%)(Table 1).
The Phytochemical investigations revealed the presence of tannins, terpenoids, cardiac glycosides and Diterpenes were predominant in aqueous and ethanol extract of Leaf and petroleum ether extract showed lesser concentration of the above components (Table 2). Similarly the aqueous, ethanol and petroleum extract of Stem showed presence of tannins, terpenoids, glycosides, alkaloids and diterpenes at various level (Table 3).
The spectrophotometric analysis carried out at 200-800 nm showed major peaks at various wavelengths. The concentration levels of test extract were standardized for analyzing the components at optimum level. The aqueous extract of leaf showed highest absorbance at 323 nm and 284 nm and ethanolic extract of leaf showed highest peak at 330nm and 202 nm (Figure 1 a & b and Table 4). This experiment showed the presence of flavonones, flavonols, chlorophylls and biflavonyls in leaf extracts. The aqueous extract of stem showed high absorbance as 0.357 at 321 nm and ethanolic extract of the same showed absorbance of 0.269, 0.075 and 0.634 at wavelength of 666 nm, 610 nm and 413 nm (Figure 2 a&b; Table 5). Petroleum ether extract of leaf showed major peaks at 669 nm, 412 nm, 283 nm, 213 nm and evidencing the presence of chlorophylls, hydrocarbons, aurones and simple phenols (Figure 3 &4; Table 6 &7). It is evidenced that the presence of prominent presence of hydroxyl cinnamic acid, chlorophylls, aurones and betacyanines. The petroleum ether extract of stem showed at 200-300 nm evidencing the possibility of presence of limited amount of terpenoids and quinines. However, the further detailed studies are essentially required using advanced equipments for the confirmation of the above compounds on the particular extracts and for their quantification.
Table 1. Physico-chemical characteristics of Leaves and Stems of Ipomoea pes-tigridis
| S. No | Physico-chemical parameters | Leaves (%) | Stems (%) |
| 1 | Loss on drying | 10.59 | 8.37 |
| 2 | Total Ash | 10.03 | 12.21 |
| 3 | Acid Insoluble ash | 0.63 | 0.04 |
| 4 | Water soluble ash | 4.80 | 7.70 |
| 5 | Alcohol soluble extractive | 6.52 | 4.57 |
| 6 | Water soluble extractive | 12.58 | 12.25 |
Table 2. Phytochemical Characteristics of Aqueous, Ethanolic and Petroleum ether extract of Ipomoeapes-tigridis Leaves
| S. No | Phytochemical Tests | I. pes-tigridis Aqueous | I. pes-tigridis Ethanol | I. pes-tigridis Petroleum ether |
| 1 | Tannins (Lead acetate test) | +++ | + | - |
| 2 | Saponnins (Foam test) | + | - | - |
| 3 | Terpenoids (Salkowsky’s test) | ++ | +++ | + |
| 4 | Cardiac glycosides (Keller-killani test) | +++ | ++ | - |
| 5 | Flavanoids (Alkaline reagent test) | - | ++ | - |
| 6 | Glycosides (Fehling’s test) | +++ | +++ | ++ |
| 7 | Anthraquinones (Modified borntrager’s) | + | + | - |
| 8 | Proteins (Millon’s test) | - | - | - |
| 9 | Alkaloids (Dragendorff’s test) |
+ |
++ |
- |
| 10 | Diterpenes | + | +++ | - |
Note: +++ Highly present ++ Medium + Trace - Not present
Table 3. Phytochemical Characteristics of Aqueous, Ethanolic and Petroleum ether extract of Ipomoeapes-tigridis Stem
| S. No | Phytochemical Tests | I. pes-tigridis Aqueous
| I. pes-tigridis Ethanol | I. pes-tigridis Petroleum ether |
| 1 | Tannins (Lead acetate test) | +++ | + | - |
| 2 | Saponnins (Foam test) | + | - | - |
| 3 | Terpenoids (Salkowsky’s test) | ++ | ++ | + |
| 4 | Cardiac glycosides (Keller-killani test) | ++ | + | - |
| 5 | Flavanoids (Alkaline reagent test) | - | + | - |
| 6 | Glycosides (Fehling’s test) | + | + | + |
| 7 | Anthraquinones (Modified borntrager’s) | + | + | - |
| 8 | Proteins (Millon’s test) | - | - | - |
| 9 | Alkaloids (Dragendorff’s test) |
+ |
+ |
- |
| 10 | Diterpenes | + | ++ | - |
Note: +++ Highly present ++ Medium + Trace - Not present
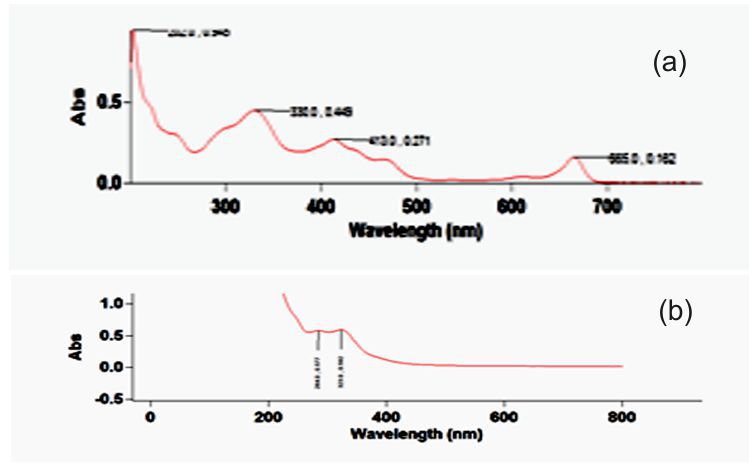
Figure 1. a & b. Spectrum of Ethanolic and Aqueous extracts of I. pes-tigridis Leaves
Table 4. Details of major peaks of the Spectrum of Ethanolic and Aqueous extracts of I. pes-tigridis leaves
| Sample Name: I. pes-tigridis leaf Ethanol 1/400 at 200-800 nm | Sample Name: I. pes-tigridis leaf Aqueous 1/160 at 200-800 nm | ||
| Wavelength | Absorbance | Wavelength | Absorbance |
| 665.0 | 0.162 | 323.0 | 0.592 |
| 413.0 | 0.271 | 284.0 | 0.577 |
| 330.0 | 0.449 | - | - |
| 202.0 | 0.945 | - | - |
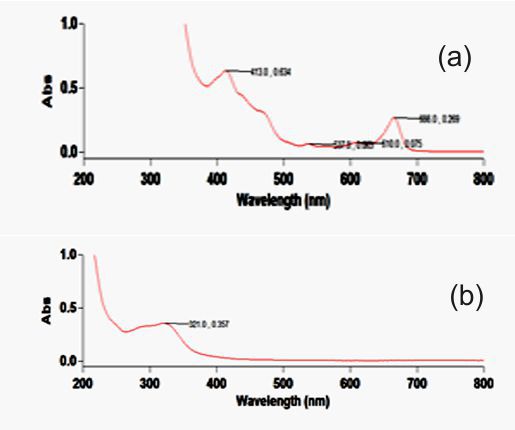
Figure 2. a & b. Spectrum of Ethanolic and Aqueous extracts of I. pes-tigridis Stem
Table 5. Details of major peaks for the Spectrum of Ethanolic and Aqueous extracts of I. pes-tigridis Stem
| Sample Name: I. pes-tigridis Stem Ethanol 1/10 at 200-800nm | Sample Name: I. pes-tigridis Stem aqueous 1/160 at 200-800 nm | ||
| Wavelength | Absorbance | Wavelength | Absorbance |
| 666.0 | 0.269 | 321.0 | 0.357 |
| 610.0 | 0.075 | - | - |
| 537.0 | 0.063 | - | - |
| 413.0 | 0.634 | - | - |
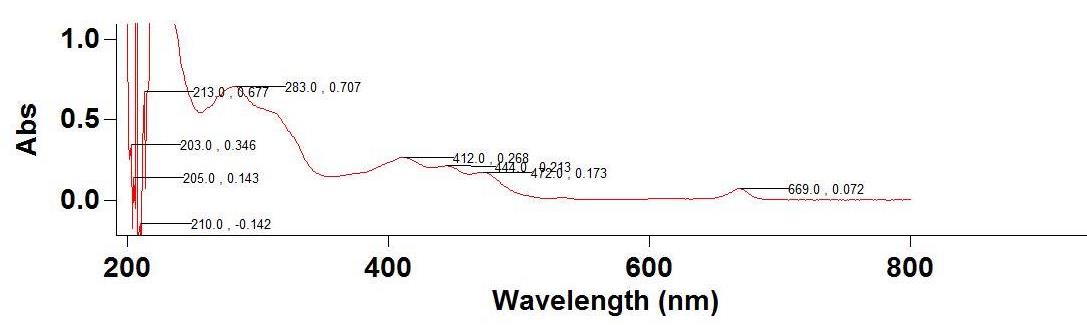
Figure 3. Spectrum of Pet. Ether extracts of I. pes-tigridis Leaves
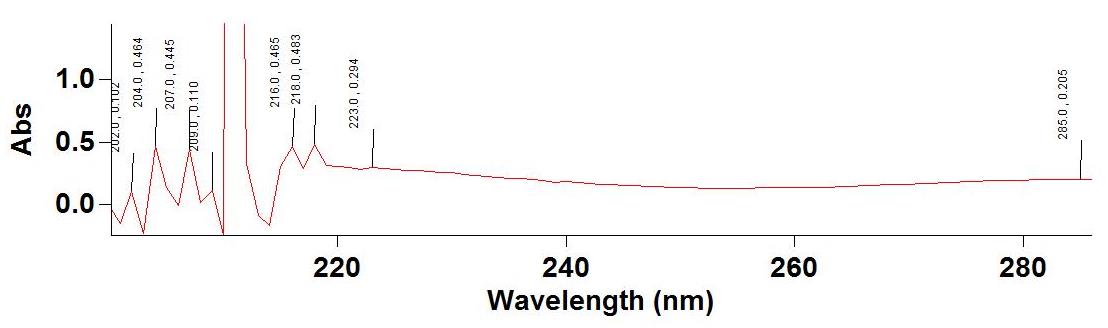
Figure 4. Spectrum of Pet. Ether extracts of I. pes-tigridis Stems
Table 6. Details of major peaks of the Spectrum of Pet. Ether extracts of I. pes-tigridis Leaf and Stem
| Sample Name: I. pes-tigridis leaf Petroleum ether1/2 at 200-800nm | Sample Name: I. pes-tigridis Stem Petroleum ether 1/20 at 200-300 nm | ||
| Wavelength | Absorbance | Wavelength | Absorbance |
| 669.0 | 0.072 | 285.0 | 0.205 |
| 472.0 | 0.173 | 223.0 | 0.294 |
| 444.0 | 0.213 | 218.0 | 0.483 |
| 412.0 | 0.268 | 216.0 | 0.465 |
| 283.0 | 0.707 | 209.0 | 0.110 |
| 213.0 | 0.677 | 207.0 | 0.445 |
| 205.0 | 0.143 | 204.0 | 0.464 |
| 203.0 | 0.346 | 202.0 | 0.102 |
Table 7. Details of Major compounds present in the different extracts of I. pes-tigridis lleaves and stems
| S. No. | Details of Plant parts and solvents | Interpretation of the major compounds based on the UV spectrum data (Harbone, 1998) |
| 1 | Aqueous extract of Leaves | Hydroxycinnamic acid, Flavonones And Flavonols |
| 2 | Ethanolic extract of Leaves | Flavonones, Flavonols& Flavones Biflavonyls, Chlorophylls, Aurones |
| 3 | Petroleum Ether extract of Leaves | Chlorophylls,Hydrocarbons &Xanthophylls, Aurones, Simple Phenols |
| 4 | Aqueous extract of Stems | Hydroxycinnamic acid |
| 5 | Ethanolic extract of Stems | Chlorophylls, Aurones, Betacyanins |
| 6. | Petroleum Ether extract of Stems | Polyacetylene |
Discussion and Conclusion
The importance of studying the medicinal values of plants in order to see the possibilities for new resources against the diseases is rising constantly all over the world. Several lead compounds in Pharma industry were revealed from the plants till now and nearly 30 % of globalpharmaceutical preparations are depending on plant resources (Shinwari and Khan, 1998; Kumar and Jayaveeran, 2014).
Ipomoea pes-tigridis is one of the potential medicinal plants under Convolvulaceae family having lot of biological properties. The present study was performed to investigate the Physico-chemical, Phytochemical and Spectroscopical characteristics of different solvent extracts of leaf and stem of I. pes-tigridis. The study showed the presence of multiple components like flavonones, flavonols, chlorophylls,biflavonyls hydrocarbons, aurones and simple phenols at varying concentrations in the aqueous, ethanol and petroleum ether extracts. The above components may contribute individually and synergistically for the medicinal value of the I. pes-tigridis. The further extensive studies in the aspects of isolation, characterization and confirmation of compounds and their mode of action at in situ are highly required.
Acknowledgement
Authors are thankful to the Director General, CCRAS, New Delhi for his support and encouragement. Authors are also expressing their gratitude to Mrs. KR. Ranjini and Mrs. KR. Neethu, Lab Technicians of Department of Biochemistry and Pathology, NRIP for their technical help.
Conflict of Interest: Nil.
References
Ali II, Umut G, Semih Y, Mehmet YD. 2012. Cytotoxicity of Aloe vera gel extracts on Allium ceparoot tip cell. Turkish Journal of Botany, 36: 263-268.
Anand T, Gokulakrishnan K. 2012. Phytochemical Analysis of Hybanthusenneaspermus using UV, FTIR and GC- MS. IOSR Journal of Pharmacy, 2(3):520-524
Begum SS, Aruna A, Sivakumar T, Premanand C, Sribhuvaneswari C. 2015. In vitro Cytotoxic Activity on Ethanolic Extracts of Leaves of Ipomoea pes-tigridis (Convolvulaceae) against Liver Hep G2 Cell line. International Journal of Ayurvedic and Herbal Medicine, 5(3):1778-1784.
Bhellum BL. 2012. Taxonomic studies on genus Ipomoea (Convolvulaceae) in the flora of Jammu and Kashmir State. Journal of Plant Biology Research.1:29-35.
Chopra RN, Nayar SL, Chopra IC. 1995. Glossary of Indian Medicinal Plants. Council of Scientific and Industrial Research, New Delhi.
Chowdhury RH, Saha R, Bhuiyan MI, Hossain MA, Kowsar SAM, Hossain MM. 2014. An In vitro Assessment of Antimicrobial, Thrombolytic and Cytotoxic Activity on Ipomoea pes-tigridis.Journal of Advancement in Medical and Life Sciences. 2(1):1-8.
Chowdhury RH, Saha R, Minhazul Islam KM, Fatema K, Afrin F, Hossain MM. 2014. Analgesic and Neuropharmacological Effect of Ethyl Acetate Extract of Ipomoea pes-tigridis in Albino Mice.European Scientific Journal, 10 (27): 344-353.
Danial FA. 1975. Typification of the new world subdivision of Ipomoea L (Convolvulaceae) Taxon 24(1):107-110.
Diego Amor-Pratsa, Harbornea JB. 1993. New sources of ergoline alkaloids within the genus Ipomoea. Biochemical Systematics and Ecology, 21(4):455-461.
Harborne JB. 1998. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. 3rd Ed. Springer Publication.
Janakiraman N, Sathish S, Johnson M. 2011. UV-Vis and FTIR Spectroscopic studies on Peristrophebicalyculata (Retz) nees. Asian Journal of Pharmaceutical and Clinical Research, Vol 4, Issue 4: 125-129
Kavit M, Patel BN and Jain BK. 2013. Phytochemical analysis of leaf extract of Phyllanthus fraternus. Research Journal of Recent Sciences. Vol 2: 12-15
Kumar GS, Jayaveeran KN. 2014. A Textbook of Pharmacognosy and Phytochemistry. S. Chand & Company, New Delhi.
Meira M, Silva EP, David JM, David JP. 2012. Review of the genus Ipomoea: Traditional uses, Chemistry and Biological Activities. Brazilian Journal of Pharmacognozy. 22(3):682-713.
Moses A, Bernard S, Oriko OR, Edward A. 2014. Preliminary Qualitative Analysis of Phytochemical Constituents of the Endemic Aloe tororoana Raynolds in Tororo, Eastern Uganda. Global Advanced Research Journal of Agricultural Science. 3(3): 096-099.
Neeli ER, Dixit VK. 2007. Ethnopharmacognostical studies on medicinal plants of Jashpur District, Chattisgarh. International Journal of Green Pharmacy. 1(1):2-4.
Pawar S, Patil DA. 2004. Observations on Folkloric Medicinal Plants of Jalgaon District, Maharashtra. Indian Journal of Traditional Knowledge. 3(4): 437-441.
Prabhu K, Karar PK, Hemalatha S, Ponnudurai K. 2011. Comparative micromorphological and Phytochemical studies on the roots of three Viburnum (Caprifoliaceae) species. Turkish Journal of Botany, 35:663-670.
Pratap GP, Sudarsanam G, Jyothi B, Prasad GP and Rasheed NMA. 2012. Pharmacognostical and analytical studies of leaves of Cardiospermumcanescens wall. International Journal of phytomedicine, 4: 386-394
Protocol for testing Ayurvedic, Siddha and Unani medicines, Dr. D.R. Lohar. 2007. Govt of India, Dept of Ayush, Pharmacopoeial Laboratory of Indian Medicines.
Ramesh R. 2010. Analgesic effects of the Aqueous Extracts of Plant Ipomoea pes-tigridis Studied in Albino Mice. Global Journal of Pharmacology,4(1):31-35.
Rao KNV, Geetha K, Alagar RM, Banji D. 2014. Quality control study and standardization of Hibiscus rosa-sinensis flowers and leaves as per WHO guidelines. Journal of Pharmacognosy and Phytochemistry, 3(4): 29-37
Reddy MN and Mishra GJ. 2012. Preliminary Phytochemical screening and Antibacterial analysis of the leaf extracts of Launaea procumbens Roxb. International Journal of Phytopharmacology, 3(2): 147-151.
Sahu PK, Gupta S. 2013. Medicinal Plants of Morning Glory: Convolvulaceae Juss. of Central India (Madhya Pradesh & Chattishgarh). Biolife. 2(2):463-469.
Santhanakrishnan D, Shankar SN and Chandrasekaran B. 2014. Studies on the phytochemistry, spectroscopic characterization and antibacterial efficacy of salicorniabrachiata. International Journal of Pharmacy and Pharmaceutical Sciences, 6:430-432.
Sathish S, Janakiraman N , Johnson M. 2012. Phytochemical Analysis of Vitex altissima L. Using UV-VIS, FTIR and GC-MS. International Journal of Pharmaceutical Sciences and Drug Research, 4(1): 56-62.
Shinwari M, Khan MA. 1998. Indigenous use of medicinal trees and shrubs of Margalla Hills National park, Islamabad. Pak J For. 48:63-90.
Silva MSH. Plantas com potancial terapiutico. 2002. http://www.prac.ufpb.br/anais.
Sofowara A. 1982. Medicinal plants & Traditional medicine in Africa; John Wiley & Sons.
Soni D, Gupta A, Solanki R, Jana GK. 2011. Pharmacognostical, phytochemical and physiochemical findings over the root extract of Hibiscus rosa-sinesis[Malvacae]. Journal of Natural Product and Plant Resources, (4): 73-79.
Suhas SN, Vilas DV. 2014. Pharmacognostic evaluation of dried leaf powder of Cardiospermum halicacabum Linn and dried seed hull powder of Hydnocarpus petandra (Buch. ham) Oken. International Journal of Research in Ayurveda and Pharmacy, 5(4): 502-507.
Thamizh Selvam N, Liji IV, Sanjaykumar YR, SanalGopi CG, Vasanth Kumar KG, Swamy GK. 2015. Evaluation of Antioxidant Activity of Averrohoa bilimbi Linn. Fruit juice in Paracetamol Intoxicated Wistar Albino Rats. Enliven: Toxicology and Allied Clinical Pharmacology, 1(1):1-7.
The Plants Database. Version 4.0.4.1996. National Plant Data Center, NRCS, USDA, Baton Rouge, LA 70874-4490. USA.
The Plants Database. Version 5.1.1.2000. National Plant Data Center, NRCS, USDA, Baton Rouge, LA 70874-4490. USA. http://plants.usda.gov.
Tiwari P, Kumar B, Kaur M, Kaur G, Kaur H. 2011. Phytochemical Screening and Extraction: A Review. Internationale Pharmaceutica Sciencia, 1 (1): 98-106.
Vinoth B, Manivasagaperumal R. 2013. Phytochemical analysis and antibacterial activity of Cardiospermum halicacabum Linn. International Journal of Current Science and Technology, 2: 009-012.
Von Rheede’s Hortus Malabaricus. 2003. English Edition. With Annotations and Modern Botanical Nomenclature By Manilal. Publication by University of Kerala, Thiruvananthapuram, 11: 217-219.