
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Pallavi Chaudhari1*, Somnath Kedar1, Shilpa Chaudhari1
1Dr. D. Y. Patil College of Pharmacy, Akurdi, Pune - 411044
*Corresponding Author
Dr. (Mrs.) Pallavi Chaudhari
Department of Pharmaceutics,
Dr. D. Y. Patil College of Pharmacy, Akurdi, Pune - 411044
Abstract
Objective: An attempt was to formulate fast dissolving tablet of Diltiazem HCl by using different synthetic superdisintegrants. Material and methods: The formulations were analyzed using Differential Scanning Calorimetry, FTIR spectroscopy. Results and conclusion: In all cases it was observed that as the concentration of superdisintegrants increased there was less disintegration time and more dissolution. Indion 414 formulation showed less disintegration time and more dissolution rate while other formulations showed more disintegration time and less dissolution rate, compared to Indion 414.
Keywords: Fast dissolving tablet, superdisintegrants, Indion 414
Introduction
Recent advance in novel drug delivery system aims to enhance the safety and efficacy of the drug molecule by formulating a dosage form being used for administration. Difficulty in swallowing is experienced by patient such as pediatric, geriatric, bedridden, disabled and mentally ill, including motion sickness and sudden episodes of allergic attacks, hence resulting in higher incidence of non-compliance and ineffective therapy. To improve the quality of life and treatment compliances of such patients fast disintegrating or orally disintegrating tablets dosage form is a better alternative for oral medication (Kuchekar et al., 2003; Yutaka et al., 2002).
The role of Superdisintegrants and their performance are of critical importance in formulation of such tablets. In more recent years, increasing attention has been paid to formulating not only fast dissolving and/or disintegrating tablets that are swallowed, but also orally disintegrating tablets that are intended to dissolve and/or disintegrate rapidly in water and to be swallowed (Seager et al., 1998).
There are many methods for formulation of rapidly disintegrating/dissolving dosage forms. Direct compression method being one of the method of the ease of preparation and cost-effectiveness. Probably one of the least recognized advantages of direct compression is the optimization of tablet disintegration, in which each primary drug particle is liberated from the tablet mass and available for dissolution (Dandagi et al., 2006; Fini et al., 2008; Hisakadzu et al., 2002).
Diltiazem hydrochloride is a BCS Class-I drug. It is very bitter and has an after taste. Diltiazem hydrochloride is a calcium channel blocker generally indicated for the treatment of angina and hypertension, and it is extensively metabolized, predominantly due to hepatic metabolism. At present, there is no fast dissolving tablet in the market; the drug is marketed as immediate sustained-release tablets, extended sustained-release capsules, and injections. The formulation of fast dissolving tablet will show rapid onset of action and avoid the hepatic metabolism.
Materials and methods
Materials
All the chemicals used in this research were of standard pharmaceutical grade. Diltiazem HCl was procured as a gift sample from Ajanta Pharma, Aurangabad. Indion 414 and Indion 234 (Ion Exchange India Ltd, Mumbai), Sodium starch glycolate, Crosscarmellose, Microcrystalline cellulose (Maple biotech Pvt Ltd, Pune), Magnesium stearate (JP Fine Chemicals, Mumbai) were of analytical reagent grade.
Methods
Fast dissolving tablets were prepared by using Diltiazem HCl with variable concentrations of superdisintegrants and other excipients. The powder blends of formulation were evaluated for bulk density, tapped density, compressibility and angle of repose using methods described earlier. Diltiazem HCl , Microcrystalline cellulose were passed from sieve no. 60 and mixed for 10 min. Superdisintegrants were then passed through sieve no.40 and added to the above mixture. Magnesium stearate, Aerosil, Aspartame was passed through sieve no.60 and added to the above mixture. The whole bulk of powder was then mixed thoroughly for 15 min. The resulting powder blends were evaluated for flow parameters. The powder blends equivalent to the 60 mg of drug were directly compressed using 8 mm concave faced round punches. Tablets were evaluated for various tablet parameters.
Pre-formulation study
Detection of Melting Point: Melting point of pure drug was determined by capillary method
Detection of absorbance maxima by UV method: In order to confirm λmax of Diltiazem HCl, 10 mg of Diltiazem HCl was weighed accurately and transferred to a 100 ml volumetric flask containing 25 ml of 0.1 N HCl; the volume was adjusted to 100 ml with 0.1 N HCl, to get a 100 µg/ml stock solution, further stock solution was diluted suitably to get 10 µg/ml solution, which was analyzed at spectrum measurement by UV-visible double beam spectrophotometer (400-200 nm) against Distilled water as a blank.
Construction of Calibration curve of Diltiazem HCl in 0.1N HCl
Preparation of standard stock solution: A standard stock solution of Diltiazem HCl of concentration 100 µg/ml was prepared by dissolving the 10mg drug in 100 ml of 0.1N HCl.
Working stock solution: The standard stock solution was then appropriately diluted with 0.1N HCl, to obtain a series of Diltiazem HCl solution in the concentration range of 1-10µg/ml. The absorbance of all the solutions was measured against blank (0.1N HCl) at237 nm using double beam spectrophotometer (Shimandzu Japan UV1700). A standard plot of absorbance v/s concentration of drug in µg was plotted. A linear graph is plotted along with linear equation.
FTIR spectrum: The FTIR spectrum was recorded on model FT-IR 8400S, Fourier Transform Infrared Spectrophotometer (Shimadzu). The pellets was preparedon KBr press using mixture of sample and KBr in about1:10 ratio. The spectrum was recorded over the wave no. range of 4000 to 400 cm (Prasant et al., 2009; Vidyavathi et al., 2009).
Drug-Excipient Compatibility Studies
Compatibility of the drug and formulation is an important pre-requisite for formulation. Therefore DSC and I.R. spectral analysis of pure drug Diltiazem HCl and physical mixture of Diltiazem HCl and superdisintegrant were carried out. IR spectra of physical mixtures (1:1) of Diltiazem HCl and various excipients, as well as the formulation were performed to find out any possible drug-excipient interaction by ATR method using FT-IR spectrophotometer.
Differential Scanning Calorimetry (DSC)
Differential scanning calorimeter (DSC) of the drugs, polymer and optimized formulation were obtained. Samples of approx. 5 mg pure drug and its physical mixtures with different excipients were hermetically sealed in flat bottomed aluminium pans and heated in the DSC instrument in an atmosphere of nitrogen to eliminate the oxidative and pyrolytic effects. The heating rate was 5ºC/min in a temperature range of 25–300ºC. The DSC thermograms were recorded.
Table 1. Composition of formulations of Diltiazem HCl fast dissolving tablets
| Formulation | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 |
| Diltiazem Hcl | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 |
| Indion 414 | 15 | - | - | - | 7.5 | - | - | 7.5 | 7.5 | - |
| Indion 234 | - | 15 | - | - | 7.5 | 7.5 | 7.5 | - | - | - |
| CCS | - | - | 15 | - | - | 7.5 | - | 7.5 | - | 7.5 |
| SSG | - | - | - | 15 | - | - | 7.5 | - | 7.5 | 7.5 |
| MCC | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 | 50 |
| Aspartame | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 |
| Aerosil | 03 | 03 | 03 | 03 | 03 | 03 | 03 | 03 | 03 | 03 |
| Mg stearate | 02 | 02 | 02 | 02 | 02 | 02 | 02 | 02 | 02 | 02 |
| Orange flavor | q.s | q.s | q.s | q.s | q.s | q.s | q.s | q.s | q.s | q.s |
| Total | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 |
Evaluation of tablets
Weight variation test
This test was performed as per I.P, 20 tablets of each formulation were weighed individually using an electronic balance (0.01mg sensitivity). The average weight was calculated and individual tablet weight was compared with the average value and the deviation was recorded. For 200 mg tablets permissible allowance is 2 out of 20 tablets can have deviation ±7.5% i.e. 15mg (Somasundaram et al., 2013).
Thickness and diameter
The thickness and diameter of the tablets was determined by using Digital Vernier caliper. Randomly 10 tablets from each batch selected & used for determination of thickness and diameter that expressed in Mean± SD and unit is mm (Somasundaram et al., 2013).
Drug content uniformity of tablets
5 tablets were weighed individually and crushed in a mortar. Quantity of powder equivalent to 60mg of Diltiazem HCl was weighed accurately and dissolved in Distilled water. The volume was made to 100ml with Distilled water. Appropriate dilutions of the resulting solutions were done. The drug contents of the resulting solutions were calculated from UV absorbance at 237 nm (Bi et al., 1996).
Hardness
Hardness values of each formulation type were determined using Monsanto hardness tester. For this the lower plunger was placed in contact with the tablet and a zero reading was taken. The upper plunger was then forced against a spring by turning a threaded bolt until the tablet fractured. The hardness was recorded from the position of the pointer (Somasundaram et al., 2013).
Friability
Friability testing was done using Roche friabilator. 20 tablets of each formulation were carefully dedusted and accurately weighed. These tablets were placed in the rotating drum of the friabilator. Drum was operated for the 100 revolutions. The tablets were removed and dedusted and reweighed. Percentage weight loss was calculated (Somasundaram et al., 2013).
Wetting time
A piece of tissue paper folded double was placed in a Petri plate (internal diameter is 6.5 cm) containing 6 ml of water. The tablet was placed on the paper and the time for complete wetting of the tablet was measured in minutes (Battu et al., 2007).
Disintegration time of tablets
The in vitro disintegration studies were carried out using Digital Tablet Disintegration Test Apparatus (scientific). One tablet was placed in each of the six tubes of the basket assembly and disk was added to each tube. This assembly was then suspended in 900ML beaker containing water maintained at 37±20C. The basket was then moved up and down through a distance of 5 to 6cm. at a frequency of 28 to 32 cycles per minutes. The time required for complete disintegration of the tablet was recorded. The test was performed for tablets of all type of formulation (Gohel et al., 2004).
Dissolution studies
The USP apparatus type-II (Veego Pvt. Ltd, India) was used for all the in vitro dissolution studies. Dissolution media used was 900ml 0.1N HCL at 50 rpm at temperature 37±0.5ᵒc. The tablets were exposed for 15 min to the dissolution medium. Appropriate aliquots (1ml sample volume) were withdrawn from the dissolution medium at predetermined intervals (0, 5, 10, 15 min. The samples were filtered through filter paper and dilute with 0.1N HCL up to 10 ml. Samples were then analyzed at λ max of 237 nm using UV/VIS double beam Spectrophotometer (Klancke et al., 2003).
Stability studies
Stability studies on the optimized formulation of fast dissolving tablet were carried out to determine the effect of temperature and humidity on the stability of the drug. The tablet (optimized batch F1) was stored in stability chamber at a 40°C±2°C/ 75%RH. The sample were withdrawn at 30 days, 90 days and subjected for visual inspection, disintegration test, drug contents and in vitro dissolution studies (Swamy et al., 2012).
Results and discussion
Melting point of Diltiazem HCl was found to be 212-214±2ͦC`. The reported value for Diltiazem HCl is ‘214ͦC’. It indicates purity of sample.
The spectrum of UV was analyzed by UV/Visible spectroscopy and λmax was found to be 237nm
Table 2. Concentration and absorbance data for Diltiazem HCl standard curve preparation in 0.1 N HCl
| Sr. No | Concentration (mcg/ml) | Absorbance (λ max – 237 nm) |
| 1 | 0 | 0 |
| 2 | 2 | 0.182 |
| 3 | 4 | 0.275 |
| 4 | 6 | 0.389 |
| 5 | 8 | 0.525 |
| 6 | 10 | 0.625 |
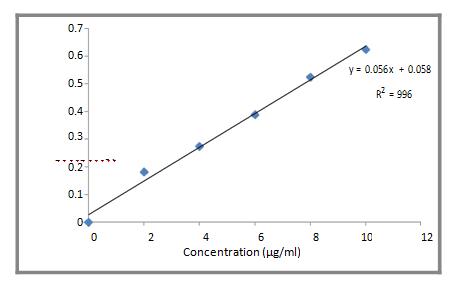
Figure 1. Calibration curve of Diltiazem HCl in 0.1 N HCl
Infrared Spectroscopic Study
The spectrum of Diltiazem HCl was recorded in ATR pellet & is shown in Fig no. .
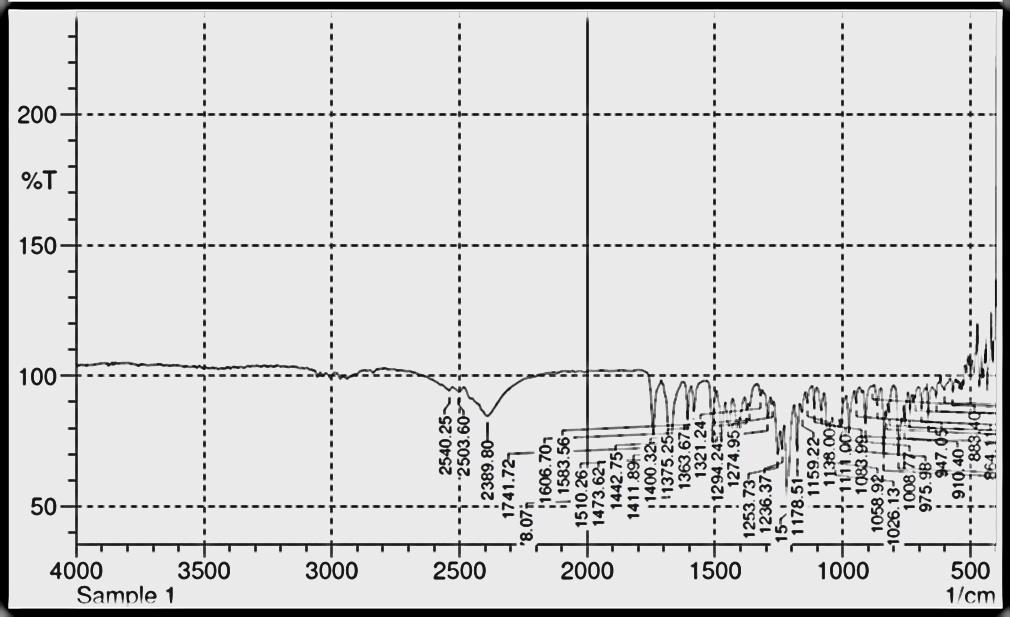
Figure 2. IR Spectra of Diltiazem HCL
Table 3. FTIR of Diltiazem HCL
| IR frequency (cm-1) | Assignment |
| 2389.8 | Amine Hcl N-H stretch |
| 1741.72 | Acetate C= O stretch |
| 1678.07 | Lactate C= O stretch |
| 839.03 | O-substituted aromatic C-H out of plane deformation |
| 781.17 | p-substituted aromatic C-H out-of plane deformation |
The DSC thermogram of the drug showed a sharp endothermic peak.
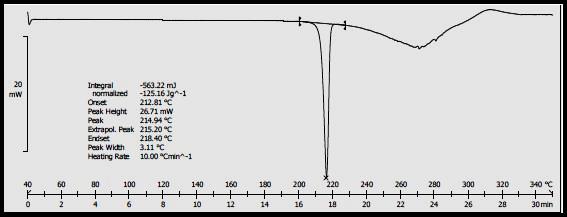
Figure 3. DSC of Diltiazem HCl
Differential scanning calorimetry of Diltiazem HCl showed the phase transition peak (sharp endothermic peak) at 214.94oC seems to be characteristic of a melting point. The melting point range is 212.81oC-215.20oC.
Table 4. Post compression parameters of formulations
| Formulation | Diameter (mm) | Thickness (mm) | Hardness (Kg/cm2) | Friability (%) | Weight variation (%) |
| F1 | 8.07±0.01 | 3.06±0.03 | 3.5±0.41 | 0.26±0.07 | 0.68±0.37 |
| F2 | 8.06±0.05 | 3.18±0.05 | 3.7±0.35 | 0.16±0.22 | 0.61±1.1 |
| F3 | 8.10±0.01 | 3.14±0.02 | 4.2±0.22 | 0.40±0.16 | 0.50±2.3 |
| F4 | 8.11±0.03 | 3.22±0.01 | 3.7±0.38 | 0.29±0.09 | 0.73±0.29 |
| F5 | 8.06±0.02 | 3.19±0.04 | 3.6±0.59 | 0.23±0.02 | 0.90±2.02 |
| F6 | 8.08±0.06 | 3.16±0.06 | 3.6±0.27 | 0.30±0.14 | 0.49±0.69 |
| F7 | 8.04±0.04 | 3.06±0.03 | 3.5±0.44 | 0.22±0.27 | 0.18±0.98 |
| F8 | 8.06±0.01 | 3.10±0.01 | 3.8±0.65 | 0.33±0.12 | 0.67±1.04 |
| F9 | 8.05±0.05 | 3.04±0.04 | 3.9±0.52 | 0.26±0.06 | 0.11±0.72 |
| F10 | 8.05±0.02 | 3.06±0.05 | 3.9±0.38 | 0.30±0.31 | 0.42±1.23 |
Table 5. Wetting Time of mouth-dissolving tablets of Diltiazem HCl
| Formulation | Time (sec) |
| F1 | 8±0.01 |
| F2 | 10±0.01 |
| F3 | 19±0.03 |
| F4 | 26±0.02 |
| F5 | 9±0.02 |
| F6 | 11±0.03 |
| F7 | 14±0.01 |
| F8 | 10±0.01 |
| F9 | 11±0.03 |
| F10 | 12±0.02 |
In vitro Disintegration Test
The disintegration time test revealed that the mouth dissolving tablet disintegrated in less than 1 min which is as per specifications given for the mouth dissolving tablets in the IP. Superdisintegrant has disintegration properties which facilitate disintegration of tablets and dissolution of drug.
The tablets were found to have disintegration time in following order Indion 414 >Indion 234>Cross carmellose sodium > sodium starch glycolate. Thus, it was found that formulation F1showed least disintegration time 13 sec.
Indion-414 showed less disintegration time than other formulation because it has more swelling capacity, easy uptake of water and rapid disintegration of tablet takes place. Indion 414 is based on a crosslinked polyacrylic acid. Tablets made with Indion 414 swells upto a very great extent about 700% because of its polymeric structure when in contact water or gastro-intestinal fluids, causing rapid disintegration without the formation of lumps.
Indion 234 when used alone showed good disintegration time but extended the duration of complete drug release. This could be due to complexation of Indion 234 with drug, as Indion 234 is also taste masking agent used for masking of bitter taste of the drug.
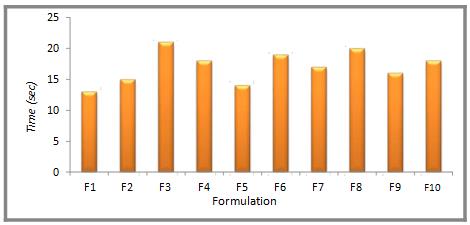
Figure 4. Disintegration time (sec) of formulations F1-F10
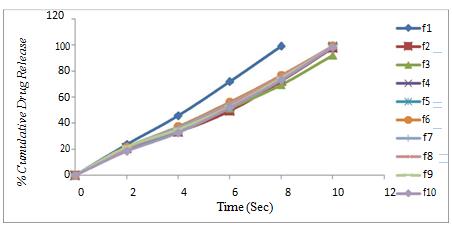
Figure 5. Drug release profile of formulation F1-F10
Dissolution Study of Formulations
The percent drug release of tablet using synthetic Superdisintegrant showed 99.31% drug release in less than 8mins. Superdisintegrant was found to increase dissolution rate. It was also seen that 10% concentration of superdisintegrants showed highest dissolution. Out of F1-F10, F1 was found to be the best formulation. The higher dissolution rate may be because of faster disintegration rate and individual entity get separated which gives maximum surface area for dissolution.
Indion 414 shows faster disintegration because of high swelling capacity than others so the dissolution of the tablets containing Indion 414 is also higher. Indion 234 when used alone showed good disintegration time but extended the duration of complete drug release. This could be due to complexation of Indion 234 with drug, as Indion 234 is also taste masking agent used for masking of bitter taste of the drug.
Stability Studies
Stability studies carried out at 40 ± 2oC/ 75± 5% RH and at room temperature for 90 days showed no significant change in colour, disintegration time, Cumulative % Drug Release and drug content of optimized formulation (F1). It indicated that formulation is stable.
Table 6. Stability study of optimized formulation F1
| Time | Appearance | Drug content | Disintegration time (sec) | Cumulative % Drug Release in 8 min. |
| Initial | White | 99.41± 2.35 | 13±0.02 | 99.31±1.47 |
| 30 days | White | 99.35± 0.70 | 12±0.02 | 99.29±1.47 |
| 90 days | White | 99.28± 1.13 | 12±0.03 | 99.27±1.47 |
Conclusion
Tablets were prepared by direct compression method. The prepared formulations were subjected to in vitro disintegration time and dissolution study. In all cases it was observed that as the concentration of superdisintegrants increases there was less disintegration time and more dissolution. Indion 414 formulation showed less disintegration time and more dissolution rate while other formulations showed more disintegration time and less dissolution rate than Indion 414 formulation. All 30 formulations were subjected to pre and post compression study from it was observed that formulation F1 shows more satisfactory results. F1 formulation was subjected to stability study which showed no change in appearance, disintegration time, dissolution time which confirmed that formulation is stable. From above discussion, it can be concluded that formulation F1 exhibited faster dissolution rate & less disintegration time which will lead to faster onset of action, improves bioavailability, improves patient compliance and satisfies all the criteria as fast dissolving tablet.
References
Battu S, Repka M, Majumdar S, Rao M. 2007. Formulation and evaluation of rapidly disintegrating tablet Fenoverine tablets: Effect of superdisintegrants. Drug Development Industrial Pharmacy, 33:1225-32.
Bi Y, Sunada H, Yonezawa Y, Danjo K, Otsuka A, Iida K. 1996. Preparation and evaluation of a compressed tablet rapidly disintegrating in the oral cavity. Chemical Pharmaceutical Bulletin, 44:2121-7.
Dandagi P, Halkatti P, Mastiholimath V, Patil M, Manvi F. 2006. Rapidly disintegrating domperidone tablets. Indian Drugs, 43(7): 594-597.
Fini A, Bergamante V, Ceschel G, Ronchi C, Fonseca D, Moraes C. 2008. Fast dispersible/slow releasing ibuprofen tablets European Journal Pharmaceutical Sciences Biopharmaceutics, 69(1):335-341.
Gohel M, Patel M, Amin A, Agrawal R, Dave R, Bariya N. 2004. Formulation design and optimization of mouth dissolve tablets of nimesulide using vacuum drying technique. AAPS Pharm Sci Tech, 5: 36.
Hisakadzu S, Yunxia B. 2002. Preparation, evaluation and optimization of rapidly disintegrating tablets. Powder Technology, 122 (2-3):188-198.
Klancke J. 2003. Dissolution testing of orally disintegrating tablets. Dissolution Technology, 10:6-8.
Kuchekar B, Badhan A, Mahajan H. 2003.Mouth Dissolving Tablets: A Novel Drug Delivery System. Pharma Times, 35: 7-9.
Lachman L, Lieberman A, Kinig JL. 1991. The Theory and Practice of Industrial Pharmacy. 4th ed. Bombay: Varghese Publishing House, 67-8.
Ratnakar R, Goswami L, Kothiyal P. 2013. Formulation and Evaluation of Fast Dissolving Tablet of Telmisartan. International Journal of Pharmaceutical and Chemical Sciences, 2(4): 2078-2085.
Seager H, 1998. Drug-delivery products and the Zydis fast-dissolving dosage form. Pharma Journal of Pharmacology, 50:375-82.
Somasundaram J, Mekonnen T. 2013. Formulation and evaluation of Diltiazem HCl oral dispersible tablets. International Journal Pharmacy Health Care Research, 1 (04):184 – 190.
Swamy N, Abbas Z. 2012. Design and Characterization of Oral Dispersible Tablets of Enalapril Maleate Using a Co-Processed Excipient. Journal of Applied Pharmaceutical Science, 2 (11):40-49.
Yutaka M, Yuki T, Masanobu Y, Ryoji T, Junko A, Kozo T. 2002.Evalution of the disintegration time of rapidly disintegrating tablets via a novel method utilizing a CCD camera. Chemical and Pharmaceutical Bulletin, 50(9): 1181-1186.